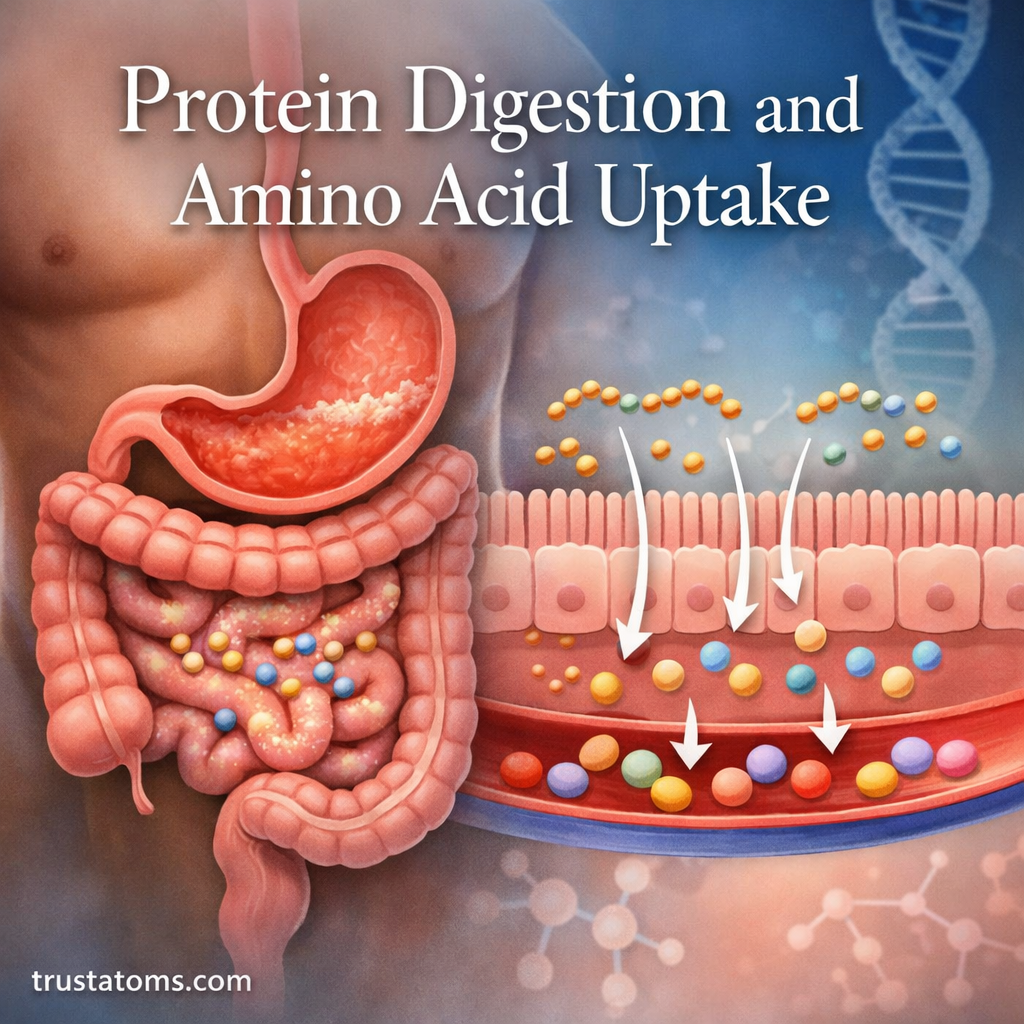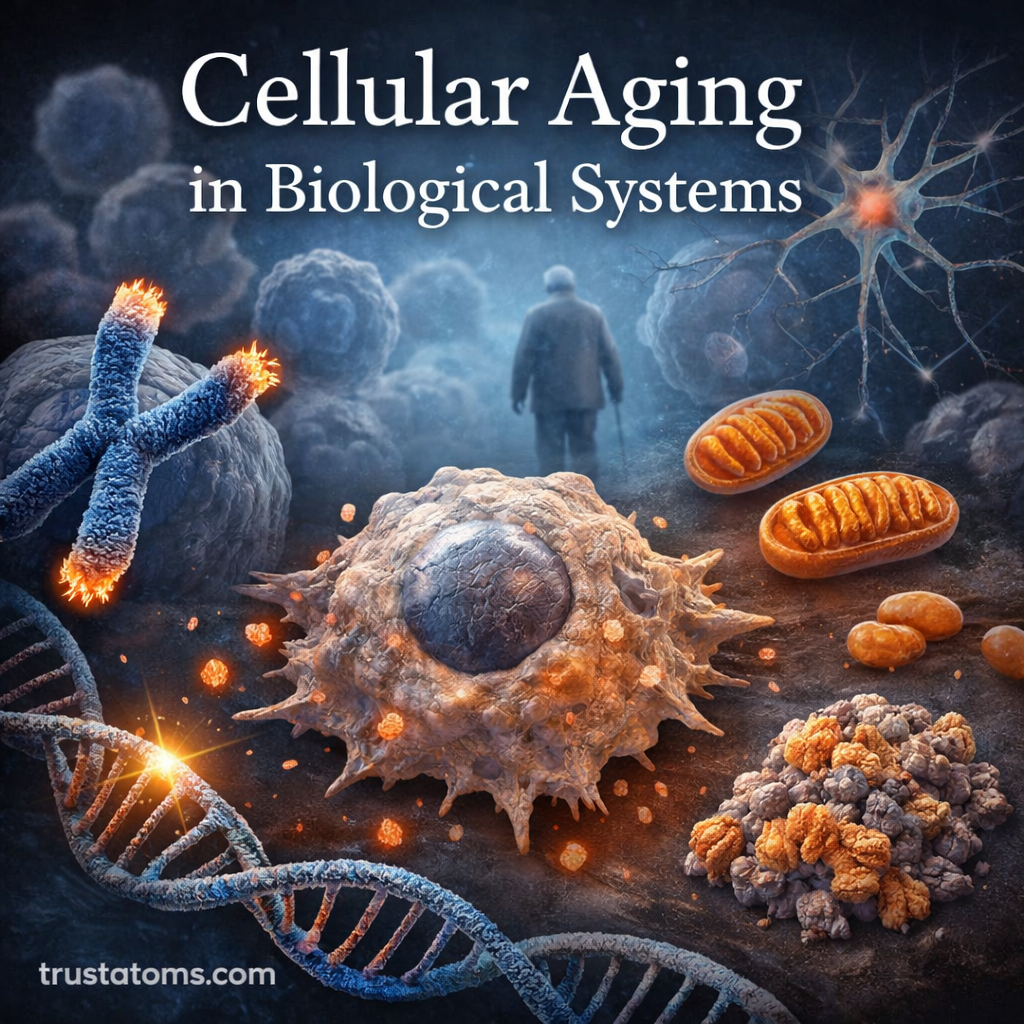
Cells are the fundamental units of life, constantly dividing, repairing, and maintaining the tissues that make up living organisms. However, cells do not function indefinitely. Over time, biological systems experience gradual changes that reduce cellular efficiency, repair capacity, and structural stability. This process is known as cellular aging.
Cellular aging plays a central role in the aging of entire organisms. From declining tissue regeneration to increased vulnerability to disease, many age-related biological changes originate at the cellular level. Understanding how and why cells age helps scientists study longevity, disease prevention, and potential therapies that may slow or modify aspects of aging.
What Is Cellular Aging?
Cellular aging refers to the progressive decline in cellular function over time. As cells divide repeatedly and encounter environmental stressors, they accumulate damage that eventually affects their ability to perform essential tasks.
Unlike organisms, cells do not simply “grow old.” Instead, aging occurs through measurable biological mechanisms that influence DNA stability, metabolic activity, and structural integrity.
Key characteristics of aging cells include:
- Reduced ability to divide
- Accumulation of molecular damage
- Declining repair mechanisms
- Changes in gene expression
- Increased susceptibility to stress
These changes gradually influence how tissues and organs function throughout an organism’s lifespan.
The Role of Cell Division and Replicative Limits
One of the most important discoveries in cellular biology is that most cells cannot divide indefinitely. This concept is known as the Hayflick limit.
The Hayflick limit describes the finite number of times a normal cell population can divide before it stops dividing. For many human cells, this limit is around 40 to 60 divisions.
This limitation occurs because of changes in chromosome structures called telomeres.
Telomeres and Replication
Telomeres are protective caps located at the ends of chromosomes. They help maintain genetic stability during cell division.
Each time a cell divides:
- Telomeres shorten slightly.
- DNA replication becomes less efficient.
- Eventually, telomeres become too short to protect the chromosome.
When telomeres reach a critically short length, the cell stops dividing and enters a state called cellular senescence.
Cellular Senescence
Cellular senescence is a state in which a cell remains alive but permanently stops dividing. Senescent cells accumulate over time in tissues as organisms age.
While senescence can serve protective purposes—such as preventing damaged cells from becoming cancerous—large numbers of senescent cells can disrupt tissue function.
Senescent cells may:
- Release inflammatory signaling molecules
- Interfere with nearby healthy cells
- Reduce tissue regeneration
- Alter normal cellular communication
The accumulation of these cells contributes to many biological features associated with aging.
DNA Damage and Repair Mechanisms
Cells constantly experience damage to their DNA. This damage can result from normal metabolic activity, environmental stressors, radiation, or chemical exposure.
Healthy cells rely on complex repair systems that detect and correct DNA errors. However, these repair systems gradually lose efficiency over time.
Common sources of DNA damage include:
- Reactive oxygen species produced during metabolism
- Ultraviolet radiation
- Environmental toxins
- Replication errors during cell division
When repair mechanisms cannot fully correct damage, mutations accumulate within the cell. Over time, this contributes to functional decline and aging-related disorders.
Oxidative Stress and Cellular Wear
Another major contributor to cellular aging is oxidative stress. Oxidative stress occurs when reactive oxygen molecules accumulate faster than the body can neutralize them.
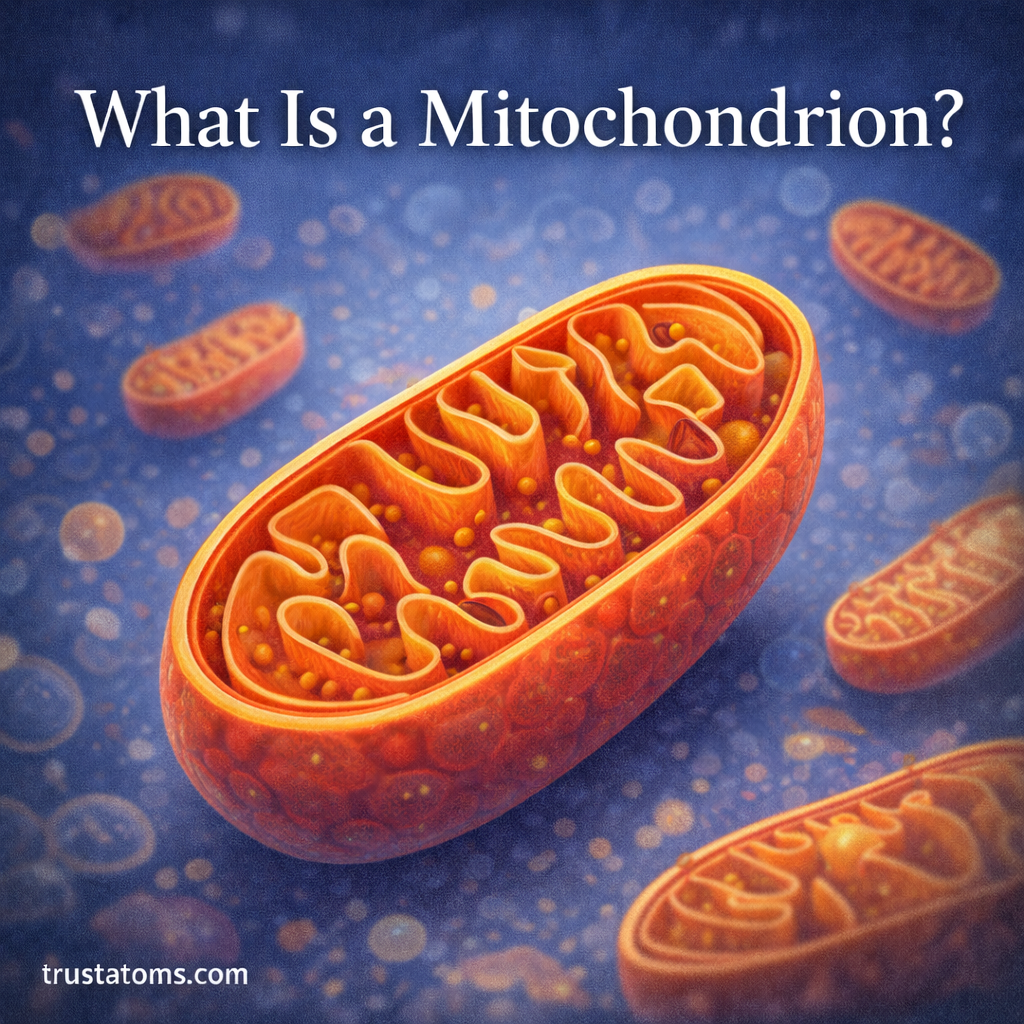
Reactive oxygen species (ROS) are natural byproducts of cellular metabolism, particularly within mitochondria—the energy-producing structures of the cell.
While small amounts of ROS are normal, excessive levels can damage:
- DNA
- Proteins
- Lipids within cell membranes
As oxidative damage accumulates, cells become less efficient at producing energy and maintaining structural stability.
Mitochondrial Decline
Mitochondria are responsible for generating most of the energy used by cells. Because they play such a central role in metabolism, mitochondrial health is closely linked to aging.
Over time, mitochondria may experience:
- Reduced energy production
- Increased generation of reactive oxygen species
- Structural damage to mitochondrial DNA
- Impaired metabolic regulation
As mitochondrial efficiency declines, cells struggle to maintain the energy required for normal biological functions.
Protein Maintenance and Cellular Quality Control
Cells rely on thousands of proteins to carry out biological tasks such as signaling, metabolism, and structural support. Maintaining protein quality is essential for proper cellular function.
Two major systems help maintain protein stability:
- Proteasome systems that break down damaged proteins
- Autophagy processes that recycle cellular components
With age, these systems gradually lose efficiency. Misfolded or damaged proteins begin to accumulate inside cells, interfering with normal biochemical activity.
Protein buildup has been associated with several age-related diseases involving cellular dysfunction.
Stem Cells and Tissue Regeneration
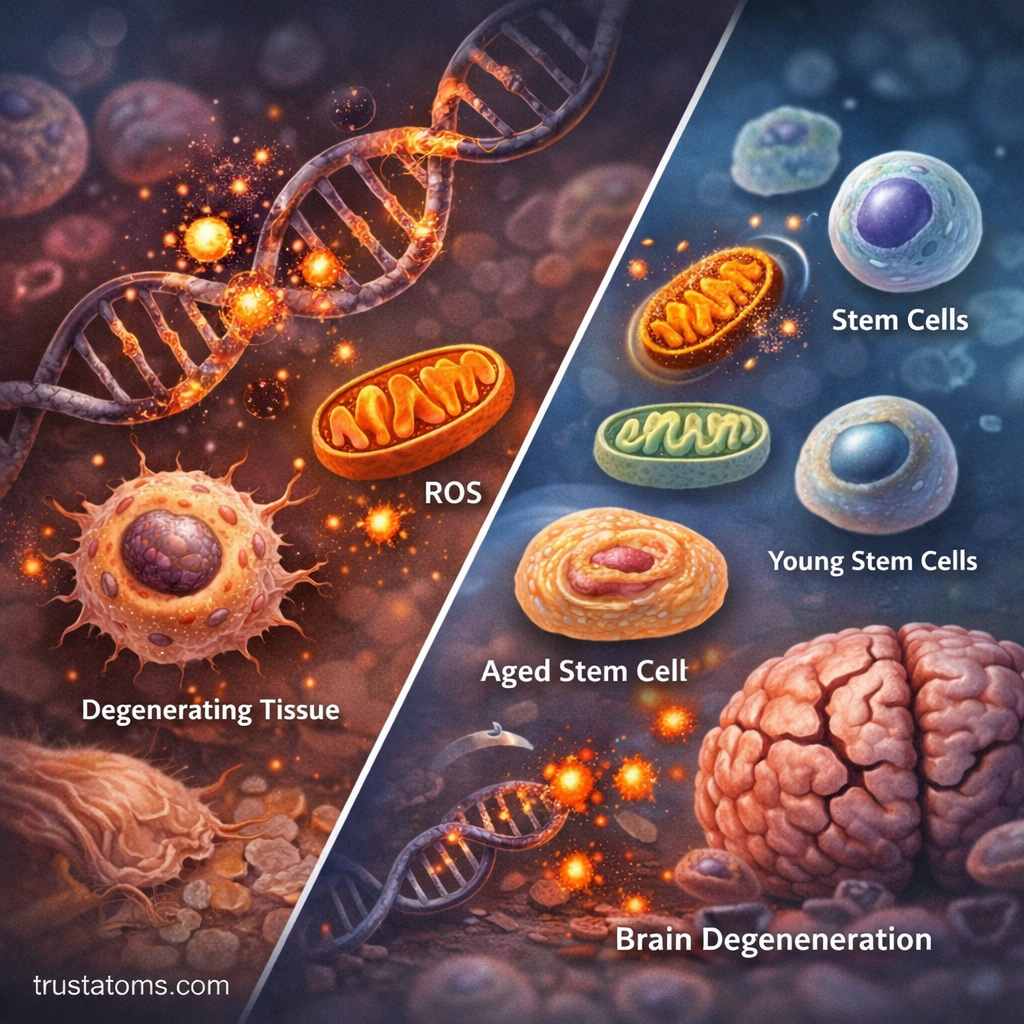
Stem cells play a critical role in maintaining tissues by replacing damaged or worn-out cells. In younger organisms, stem cells actively divide to support tissue renewal.
However, stem cell activity declines with age.
This decline leads to:
- Slower tissue repair
- Reduced regeneration after injury
- Decreased immune system responsiveness
- Loss of muscle and organ resilience
Stem cell aging is one reason why healing and recovery often become slower later in life.
Cellular Communication Changes
Cells communicate through chemical signals that coordinate biological processes across tissues and organs.
As cells age, these signaling systems may become less precise. This disruption can lead to altered immune responses, chronic low-level inflammation, and impaired coordination between different biological systems.
Scientists sometimes refer to this phenomenon as “inflammaging,” which describes the gradual increase in inflammatory signaling that occurs during aging.
Factors That Influence Cellular Aging
Although cellular aging is a natural biological process, several factors can influence its speed and impact.
Genetic Factors
Some individuals inherit genes that affect how efficiently their cells repair damage, maintain telomeres, or regulate metabolism.
Environmental Influences
External factors can accelerate cellular stress. These may include:
- Exposure to pollutants
- Radiation
- Chronic stress
- Poor nutrition
Lifestyle Factors
Certain lifestyle choices may influence cellular health over time, including:
- Regular physical activity
- Balanced nutrition
- Adequate sleep
- Reduced exposure to toxins
While lifestyle cannot stop cellular aging, it can influence how cells respond to stress and damage.
Cellular Aging and Age-Related Diseases
Many age-related conditions are closely linked to cellular aging processes. As cells lose efficiency and accumulate damage, tissues may become more vulnerable to dysfunction.
Examples of diseases associated with cellular aging include:
- Neurodegenerative disorders
- Cardiovascular disease
- Metabolic disorders
- Certain cancers
Researchers continue to investigate how slowing cellular damage or improving repair mechanisms might influence long-term health.
Why Cellular Aging Matters in Biology
Cellular aging is one of the most important topics in modern biology because it connects molecular processes with large-scale biological outcomes.
Studying cellular aging helps scientists:
- Understand how organisms age over time
- Identify biological mechanisms behind age-related diseases
- Explore strategies that support cellular health
- Investigate longevity and regenerative medicine
Advances in cellular biology continue to reveal how complex and interconnected aging processes are across biological systems.
Final Thoughts
Cellular aging is not caused by a single factor but by a combination of biological processes that gradually influence cellular structure, repair capacity, and communication.
Telomere shortening, DNA damage, oxidative stress, mitochondrial decline, and protein accumulation all contribute to the gradual decline of cellular function. As these changes accumulate across millions of cells, they shape how tissues and organs age throughout an organism’s life.
By studying cellular aging, scientists gain deeper insight into how life changes over time—and how biological systems maintain balance despite constant molecular challenges.