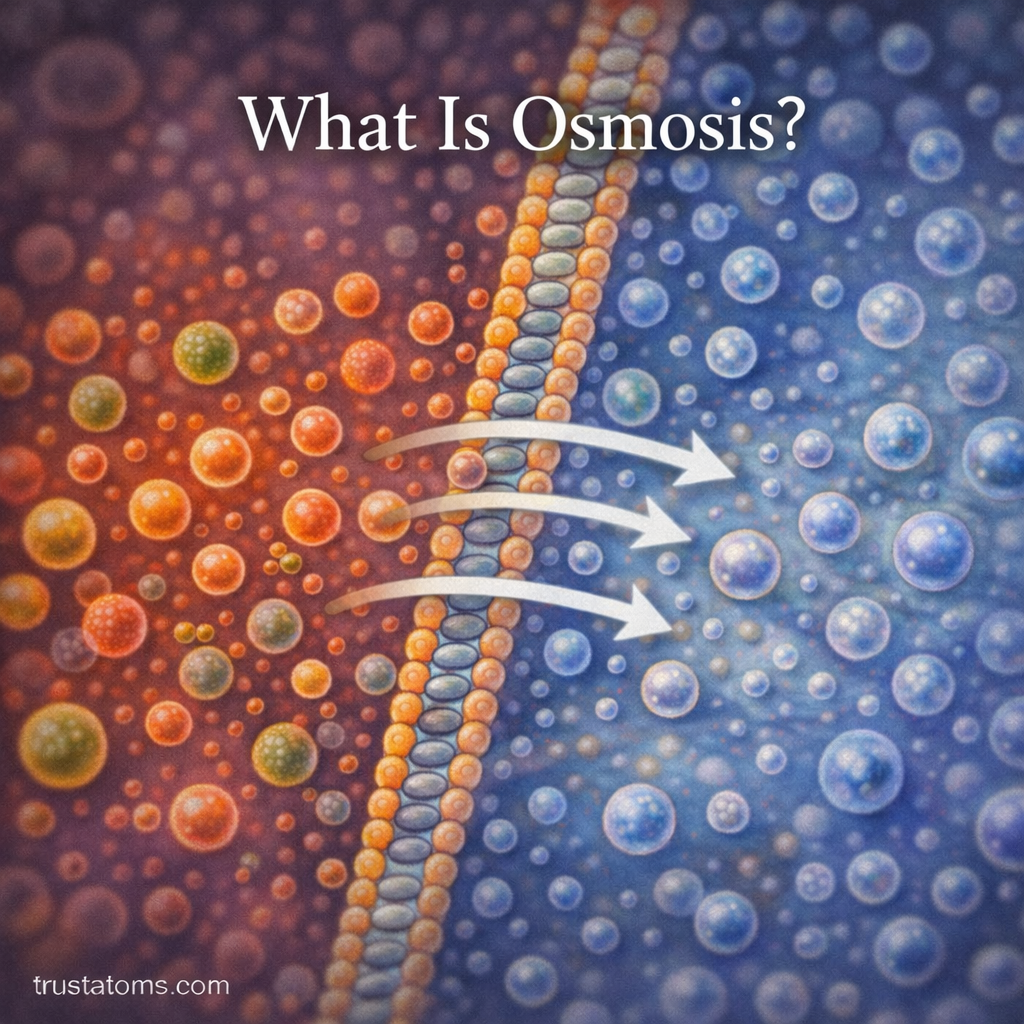
Osmosis is a fundamental biological process that helps regulate the movement of water across cell membranes. In living organisms, maintaining the right balance of water inside and outside cells is essential for survival. Osmosis allows cells to absorb or release water naturally, helping maintain internal stability.
This process occurs across a selectively permeable membrane, meaning the membrane allows some substances to pass through while blocking others. Water moves through the membrane from an area of lower solute concentration to an area of higher solute concentration.
Osmosis plays a critical role in cell hydration, nutrient transport, and maintaining proper cellular pressure in both plant and animal cells.
The Basic Definition of Osmosis
Osmosis is the passive movement of water molecules across a selectively permeable membrane from a region of higher water concentration to a region of lower water concentration.
Because solutes reduce the concentration of free water molecules, water moves toward areas where solute concentration is higher.
Key characteristics of osmosis include:
- Movement of water molecules only
- Occurs across a selectively permeable membrane
- Does not require cellular energy
- Continues until equilibrium is reached
At equilibrium, water still moves in both directions, but the overall concentration on each side of the membrane remains stable.
How Osmosis Works at the Molecular Level
Water molecules are constantly moving due to thermal energy. When a membrane separates two solutions with different solute concentrations, water molecules diffuse across the membrane.
This movement occurs because:
- Water molecules collide with the membrane.
- The membrane allows water to pass through small channels.
- Water flows toward the side with higher solute concentration.
As water moves across the membrane, the concentration difference gradually decreases until balance is achieved.
The Role of Selectively Permeable Membranes
A selectively permeable membrane allows certain molecules to pass through while blocking others.
Cell membranes are made primarily of a phospholipid bilayer, which acts as a barrier that controls molecular movement.
Water molecules can pass through the membrane by:
- Diffusing directly through the membrane
- Moving through specialized proteins called aquaporins
Aquaporins are channels that allow water to cross the membrane more efficiently.
Because many dissolved substances cannot pass through the membrane easily, water movement becomes the primary way to balance concentrations.
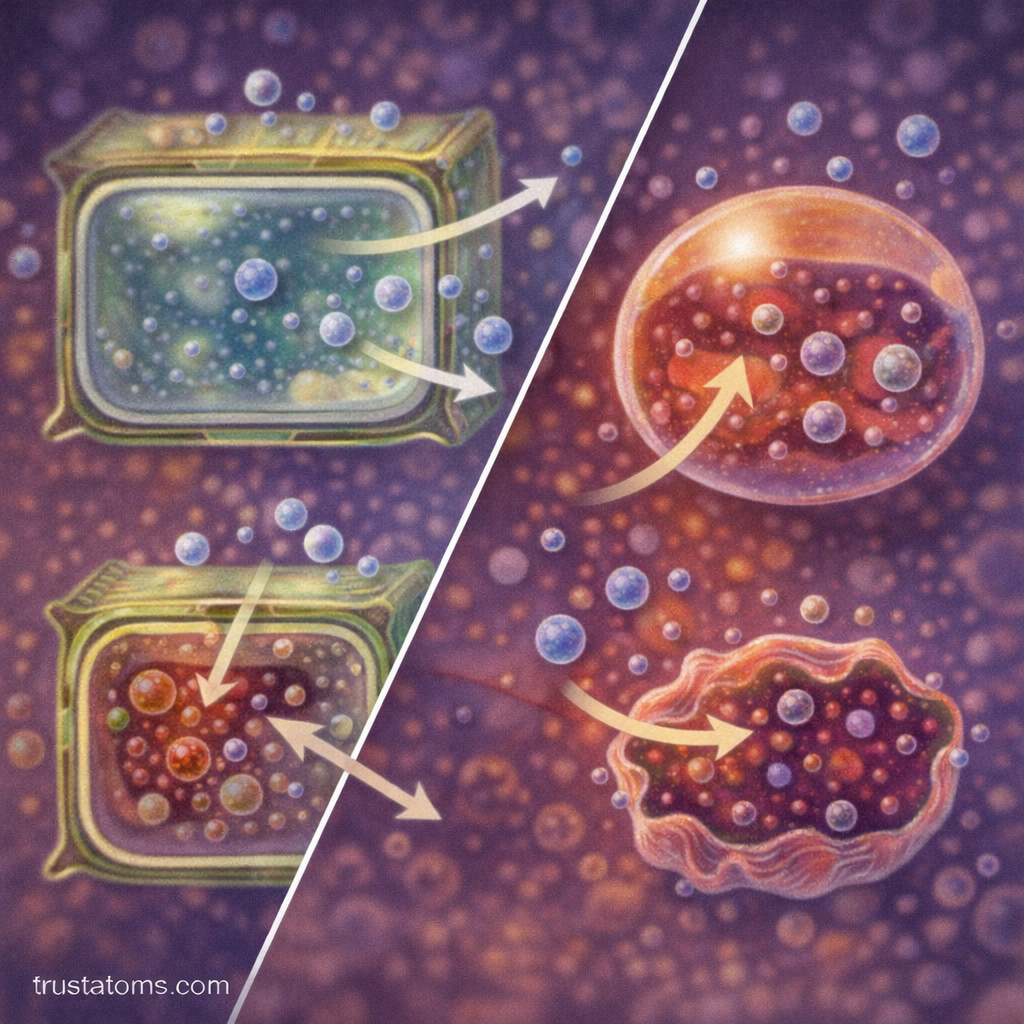
Osmotic Solutions and Their Effects on Cells
The direction of water movement during osmosis depends on the concentration of solutes in the surrounding environment. These environments are commonly described using three terms.
Hypotonic Solution
A hypotonic solution contains a lower concentration of solutes compared to the inside of the cell.
In this environment:
- Water moves into the cell.
- The cell may swell as water enters.
- Excessive swelling can cause some cells to rupture.
Hypertonic Solution
A hypertonic solution contains a higher concentration of solutes compared to the inside of the cell.
In this environment:
- Water moves out of the cell.
- The cell shrinks as it loses water.
Isotonic Solution
An isotonic solution contains equal solute concentrations inside and outside the cell.
In this environment:
- Water moves in both directions equally.
- The cell maintains its normal size.
These conditions help explain how cells respond to different external environments.
Osmosis in Plant Cells
Osmosis plays a particularly important role in plant cells because it helps maintain internal pressure known as turgor pressure.
When plant cells absorb water:
- The central vacuole fills with water.
- The cell membrane pushes against the cell wall.
- The plant cell becomes firm and stable.
This pressure helps support plant structure and prevents wilting.
If plant cells lose too much water in a hypertonic environment, they may shrink and pull away from the cell wall in a process called plasmolysis.
Osmosis in Animal Cells
Animal cells lack rigid cell walls, so their response to osmotic changes differs from plant cells.
In animal cells:
- Hypotonic environments may cause cells to swell.
- Hypertonic environments cause cells to shrink.
- Isotonic conditions help maintain normal cell shape.
Because of these differences, many biological systems carefully regulate the osmotic balance of bodily fluids.
Osmosis in Biological Systems
Osmosis is involved in many processes throughout living organisms.
Examples include:
- Water absorption in plant roots
- Fluid balance in human cells
- Nutrient transport in tissues
- Regulation of blood cell volume
In humans and other animals, organs such as the kidneys help regulate water balance by controlling the concentration of fluids in the body.
Osmosis Compared With Diffusion
Osmosis is closely related to diffusion, but there is an important difference.
Diffusion refers to the movement of any molecules from high concentration to low concentration.
Osmosis specifically refers to the movement of water molecules across a selectively permeable membrane.
Key differences include:
Diffusion:
- Can involve many types of molecules
- May occur with or without a membrane
- Moves substances down their concentration gradient
Osmosis:
- Involves only water molecules
- Requires a selectively permeable membrane
- Moves water toward higher solute concentration
Both processes are examples of passive transport because they do not require energy from the cell.
Why Osmosis Matters in Biology
Osmosis is essential for maintaining cellular stability and proper fluid balance. By regulating the movement of water, cells can maintain the conditions necessary for metabolic processes.
Without osmosis, cells would struggle to:
- Maintain proper hydration
- Transport dissolved substances
- Regulate internal pressure
- Balance chemical concentrations
Because of its importance, osmosis is a core concept in biology and physiology.
Final Thoughts
Osmosis is a simple yet essential process that governs the movement of water across cellular membranes. By allowing water to move naturally toward areas of higher solute concentration, cells maintain balance and support essential biological functions.
From maintaining plant structure to regulating fluid balance in animal cells, osmosis plays a critical role in sustaining life. Understanding this process helps explain how cells interact with their environment and maintain stable internal conditions.