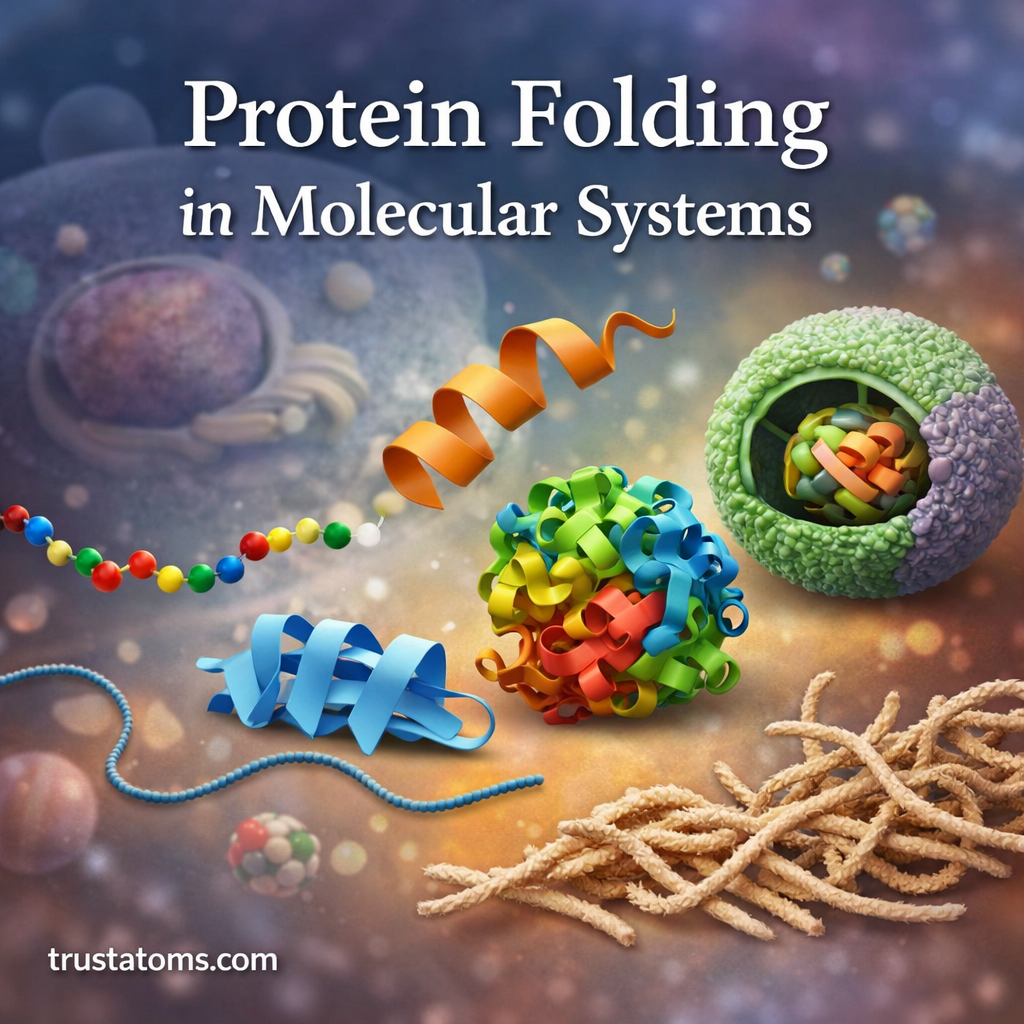
Proteins are essential biological molecules responsible for countless processes inside living cells. From catalyzing chemical reactions to transporting oxygen and supporting cellular structure, proteins perform their functions based largely on their shape.
Protein folding is the biological process in which a newly synthesized chain of amino acids folds into a specific three-dimensional structure. This folding process is critical because the function of a protein depends on its final shape.
In molecular systems, protein folding is guided by chemical interactions between amino acids, environmental conditions within the cell, and specialized helper molecules. Understanding protein folding helps scientists study diseases, drug design, and the fundamental mechanics of life itself.
What Is Protein Folding?
Protein folding refers to the process by which a linear sequence of amino acids transforms into a stable three-dimensional structure.
Proteins begin as long chains formed during protein synthesis in ribosomes. These chains are called polypeptides. While they start as flexible strands, they quickly fold into complex structures driven by molecular forces.
The folding process is not random. Instead, it follows rules determined by the chemical properties of amino acids and the environment surrounding the protein.
Key characteristics of protein folding include:
- Formation of stable structural patterns
- Minimization of molecular energy
- Specific interactions between amino acid side chains
- Rapid folding within milliseconds to seconds
When folding occurs correctly, the protein becomes functional. If folding fails, the protein may lose its function or form harmful aggregates.
Levels of Protein Structure
Protein folding organizes amino acids into multiple structural levels. Each level builds upon the previous one to create the final functional protein.
Primary Structure
The primary structure is the linear sequence of amino acids connected by peptide bonds.
Important points about primary structure:
- Determined by genetic information in DNA
- Specifies the order of amino acids in the protein chain
- Directly influences how the protein will fold
Even a single change in this sequence can significantly affect protein folding and function.
Secondary Structure
Secondary structures arise from hydrogen bonding between atoms in the protein backbone.
Common secondary structures include:
- Alpha helices – spiral-shaped structures stabilized by hydrogen bonds
- Beta sheets – folded sheets formed by parallel or antiparallel strands
These patterns provide local stability and serve as building blocks for larger structures.
Tertiary Structure
The tertiary structure represents the overall three-dimensional shape of a single protein molecule.
This structure forms through interactions between amino acid side chains, including:
- Hydrophobic interactions
- Hydrogen bonds
- Ionic interactions
- Disulfide bridges
The tertiary structure determines the protein’s biological activity.
Quaternary Structure
Some proteins consist of multiple polypeptide chains. When these chains assemble into a functional complex, they form a quaternary structure.
Examples include:
- Hemoglobin (four protein subunits)
- DNA polymerase complexes
- Structural protein assemblies
These multi-unit proteins rely on coordinated folding and assembly to function properly.
Molecular Forces That Drive Protein Folding
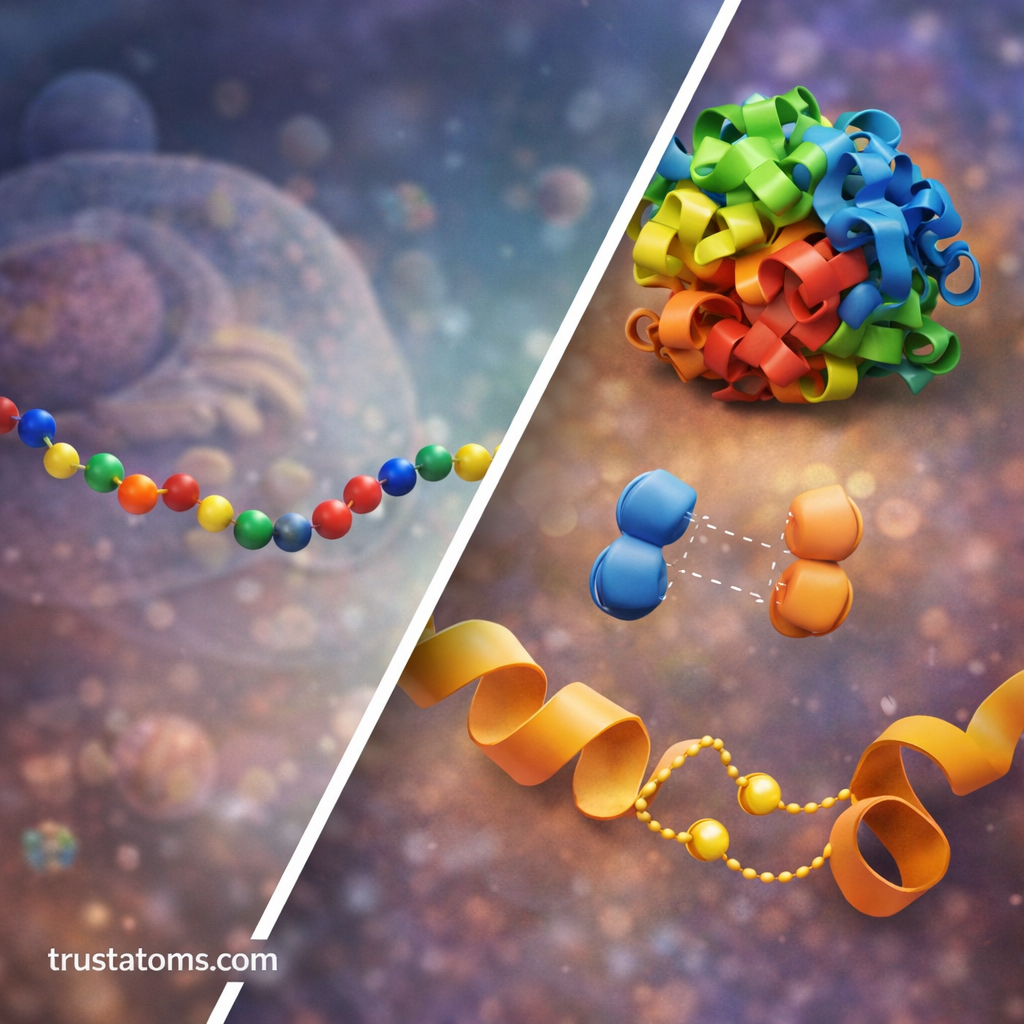
Protein folding is guided by physical and chemical interactions between atoms in the polypeptide chain and the surrounding environment.
Major forces involved include:
Hydrophobic Interactions
Many amino acids contain hydrophobic (water-repelling) side chains.
During folding:
- Hydrophobic residues tend to cluster inside the protein
- Hydrophilic residues remain exposed to water
This effect helps drive the protein into a compact shape.
Hydrogen Bonding
Hydrogen bonds stabilize many protein structures by linking different parts of the chain.
They help form:
- Alpha helices
- Beta sheets
- Internal stabilizing connections
Although individually weak, large numbers of hydrogen bonds significantly stabilize proteins.
Ionic Interactions
Charged amino acids can attract or repel each other.
These electrostatic interactions help shape the folding process and maintain stability under physiological conditions.
Disulfide Bonds
Some proteins contain covalent bonds between cysteine residues, known as disulfide bridges.
These bonds:
- Provide strong structural stability
- Often occur in extracellular proteins
- Help maintain folded shapes in harsh environments
The Protein Folding Pathway
Proteins generally fold through a series of intermediate states rather than forming their final structure instantly.
A simplified folding pathway may include:
- Formation of local secondary structures
- Collapse into a partially folded intermediate
- Rearrangement of structural elements
- Stabilization into the final native structure
Scientists sometimes describe this process using the concept of an energy landscape, where proteins move toward their lowest-energy, most stable configuration.
Molecular Chaperones and Assisted Folding
While some proteins fold spontaneously, many require assistance from specialized proteins known as molecular chaperones.
Chaperones help by:
- Preventing incorrect folding
- Protecting partially folded proteins from aggregation
- Providing isolated environments for proper folding
Examples of molecular chaperone systems include:
- Heat shock proteins (HSPs)
- Chaperonin complexes
- Folding catalysts such as protein disulfide isomerase
These systems are especially important in crowded cellular environments where misfolding risks increase.
Protein Misfolding and Disease
When proteins fail to fold correctly, they can become unstable or form harmful aggregates.
Protein misfolding is associated with several diseases, including:
- Alzheimer’s disease
- Parkinson’s disease
- Huntington’s disease
- Prion diseases
In many cases, misfolded proteins accumulate into structures called amyloid fibrils, which interfere with normal cellular functions.
Understanding protein folding mechanisms allows scientists to study how these diseases develop and explore possible treatments.
Why Protein Folding Matters in Biology and Medicine
Protein folding plays a central role in nearly every biological process.
Its importance extends to several scientific fields:
Medicine
Understanding folding helps researchers:
- Design targeted drugs
- Develop treatments for protein misfolding diseases
- Engineer therapeutic proteins
Biotechnology
Protein folding knowledge is used to:
- Produce functional enzymes in laboratories
- Design synthetic proteins
- Improve industrial biocatalysts
Structural Biology
Advanced technologies such as cryo-electron microscopy and computational modeling allow scientists to analyze protein structures with remarkable precision.
These methods deepen our understanding of how molecular systems operate.
Final Thoughts
Protein folding is one of the most fundamental processes in molecular biology. Through a series of chemical interactions and structural transitions, simple chains of amino acids transform into complex three-dimensional machines that power life.
From cellular metabolism to immune responses, the function of proteins depends entirely on their ability to fold into the correct shape. Continued research into protein folding helps scientists understand disease mechanisms, improve drug development, and uncover the intricate molecular systems that sustain living organisms.