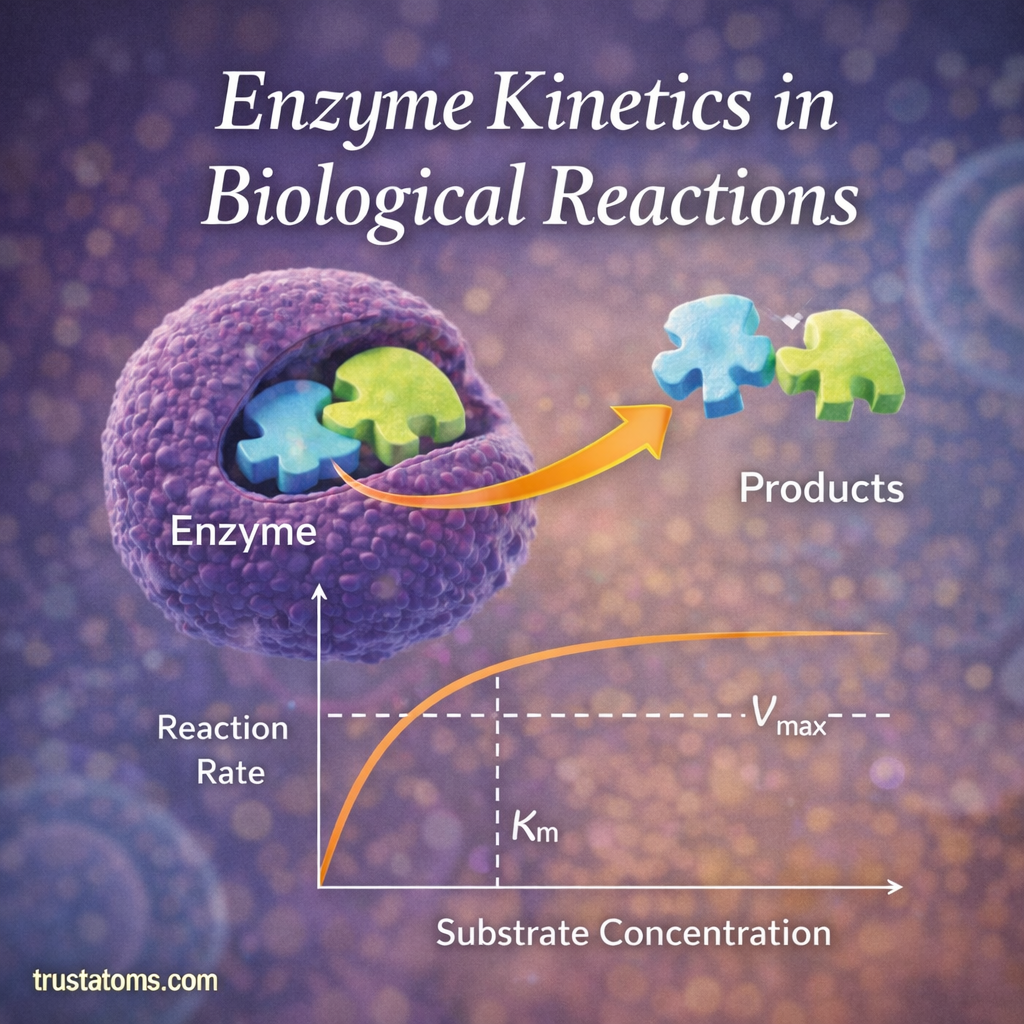
Enzyme kinetics is the study of how quickly enzymes catalyze chemical reactions and how different factors influence those reaction rates. Because enzymes control nearly every biochemical process in living organisms, understanding enzyme kinetics is essential for studying metabolism, drug development, and molecular biology.
In biological systems, enzymes accelerate reactions by lowering the activation energy required for molecules to interact. Enzyme kinetics focuses on measuring how fast these reactions occur and identifying the conditions that affect enzyme efficiency.
Scientists analyze enzyme kinetics to understand how enzymes behave in cells and how biological reactions are regulated.
What Is Enzyme Kinetics?
Enzyme kinetics refers to the quantitative study of enzyme-catalyzed reaction rates.
Researchers investigate questions such as:
- How quickly an enzyme converts substrates into products
- How substrate concentration influences reaction speed
- How environmental factors affect enzyme activity
- How inhibitors alter enzyme performance
By measuring these variables, scientists can determine how enzymes function under different biological conditions.
Basic Components of Enzyme Reactions
Before exploring enzyme kinetics, it helps to understand the main participants in enzyme-driven reactions.
Enzyme
An enzyme is a biological catalyst that accelerates chemical reactions without being consumed.
Substrate
The substrate is the molecule that the enzyme acts upon. It binds to the enzyme’s active site where the reaction occurs.
Enzyme–Substrate Complex
When the substrate binds to the enzyme, an enzyme–substrate complex forms. This temporary structure allows the chemical reaction to proceed efficiently.
Product
The product is the molecule produced after the reaction is complete. Once released, the enzyme can catalyze another reaction.
The overall reaction can be simplified as:
Enzyme + Substrate → Enzyme–Substrate Complex → Enzyme + Product
Reaction Rate in Enzyme Kinetics
The reaction rate refers to how quickly substrates are converted into products.
Reaction rates are usually measured by observing:
- The appearance of product over time
- The disappearance of substrate over time
In enzyme studies, the initial reaction rate is often measured because it reflects enzyme activity before the reaction slows down.
Factors influencing reaction rate include:
- Substrate concentration
- Enzyme concentration
- Temperature
- pH
- Presence of inhibitors or activators
The Michaelis–Menten Model
One of the most widely used models for enzyme kinetics is the Michaelis–Menten equation.
This model describes how reaction rate changes as substrate concentration increases.
The Michaelis–Menten model helps scientists understand two important kinetic parameters:
Vmax
Vmax represents the maximum reaction rate when all enzyme molecules are saturated with substrate.
At this point:
- Every enzyme active site is occupied.
- Increasing substrate concentration no longer increases the reaction rate.
Km (Michaelis Constant)
The Michaelis constant (Km) represents the substrate concentration at which the reaction rate reaches half of Vmax.
Km provides insight into enzyme affinity for the substrate:
- Low Km indicates strong enzyme–substrate binding.
- High Km indicates weaker binding.
These parameters help researchers compare enzyme efficiency across biological systems.
The Enzyme Saturation Effect
At low substrate concentrations:
- Reaction rates increase rapidly as substrate levels rise.
At higher concentrations:
- Enzymes become saturated with substrate.
- Reaction rates level off and approach Vmax.
This saturation occurs because there are only a limited number of active sites available on enzyme molecules.
Once all active sites are occupied, adding more substrate cannot further increase the reaction speed.
Factors That Affect Enzyme Kinetics
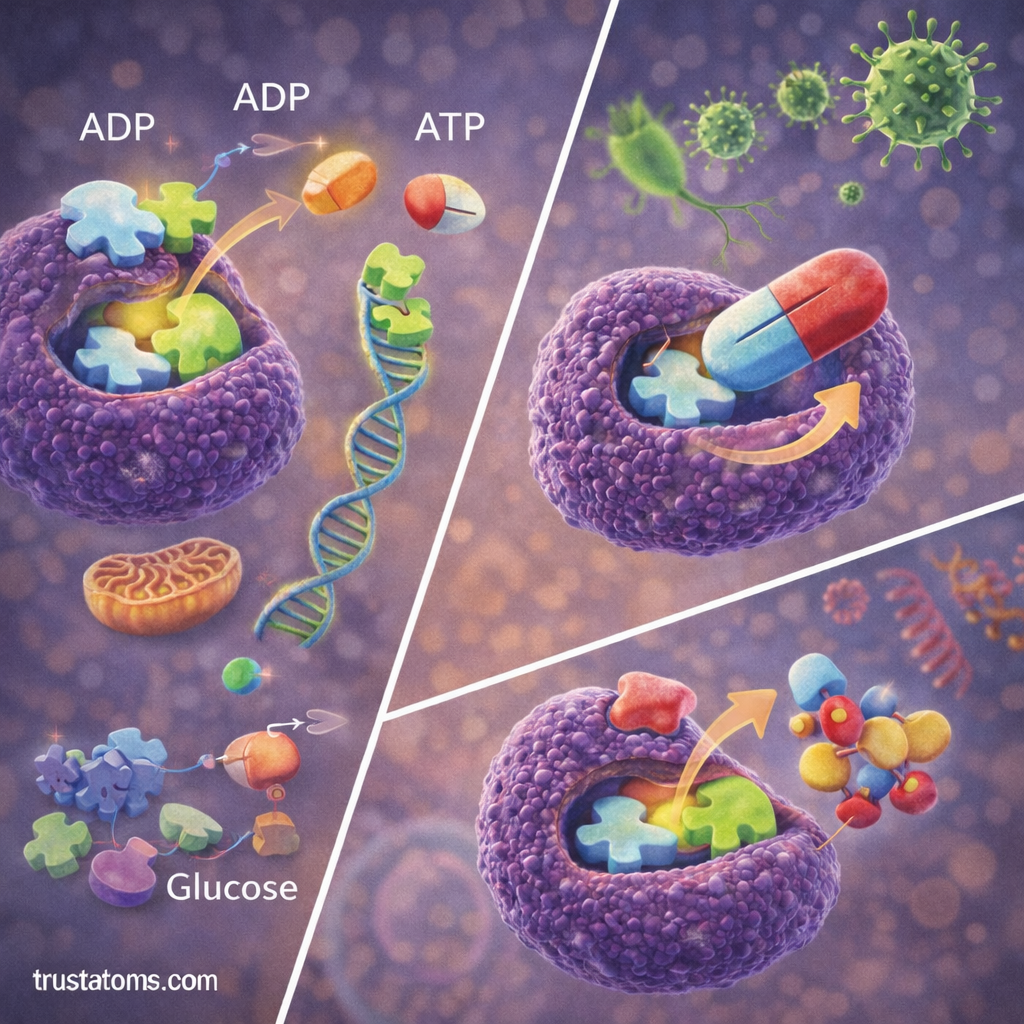
Several environmental and biological conditions influence enzyme reaction rates.
Substrate Concentration
As substrate concentration increases:
- Reaction rates initially increase quickly.
- Eventually, the rate reaches a plateau when enzymes become saturated.
Enzyme Concentration
Increasing enzyme concentration typically increases the reaction rate because more active sites become available for substrate binding.
If substrate is abundant, doubling enzyme concentration can roughly double the reaction rate.
Temperature
Temperature influences enzyme activity by affecting molecular motion.
Effects include:
- Higher temperatures increase reaction speed.
- Extremely high temperatures may cause enzyme denaturation.
Most enzymes in the human body function best near 37°C.
pH Levels
Each enzyme has an optimal pH range where it functions best.
Examples include:
- Pepsin working best in acidic stomach conditions
- Trypsin functioning in the alkaline environment of the small intestine
Outside the optimal pH range, enzyme structure may change and reduce activity.
Cofactors and Coenzymes
Some enzymes require additional molecules to function properly.
These helpers include:
- Cofactors – inorganic molecules such as metal ions
- Coenzymes – organic molecules such as vitamins or vitamin-derived compounds
Cofactors assist enzymes in stabilizing substrates or facilitating chemical reactions.
Enzyme Inhibition and Kinetics
Enzyme kinetics also examines how inhibitors affect reaction rates.
Inhibitors are molecules that reduce or prevent enzyme activity.
Competitive Inhibition
In competitive inhibition:
- The inhibitor resembles the substrate.
- It competes for binding at the enzyme’s active site.
This type of inhibition can often be overcome by increasing substrate concentration.
Noncompetitive Inhibition
In noncompetitive inhibition:
- The inhibitor binds to a different site on the enzyme.
- This alters the enzyme’s shape and reduces activity.
Because the active site changes shape, adding more substrate does not restore the reaction rate.
Why Enzyme Kinetics Matters in Biology
Studying enzyme kinetics helps scientists understand how biological systems regulate chemical reactions.
Important applications include:
- Drug development – identifying molecules that inhibit disease-related enzymes
- Metabolic research – studying biochemical pathways in cells
- Biotechnology – improving industrial enzymes used in manufacturing
- Medical diagnostics – analyzing enzyme activity to detect disease
By analyzing enzyme kinetics, researchers gain insight into how enzymes function under normal and abnormal biological conditions.
Enzyme Kinetics in Medicine and Biotechnology
Enzyme kinetics plays a crucial role in modern science and medicine.
Examples of its applications include:
- Designing drugs that inhibit viral or bacterial enzymes
- Studying enzyme deficiencies in genetic disorders
- Developing enzymes for food production and industrial processes
- Understanding metabolic diseases such as diabetes
Pharmaceutical scientists often use enzyme kinetics to determine how effectively drugs bind to enzyme targets.
Final Thoughts
Enzyme kinetics provides a powerful framework for understanding how enzymes drive the chemical reactions necessary for life. By studying reaction rates, substrate interactions, and environmental influences, scientists can uncover the mechanisms that control metabolism and cellular function.
From medical research to biotechnology, enzyme kinetics continues to be a central tool for exploring biological systems and developing new treatments for disease.