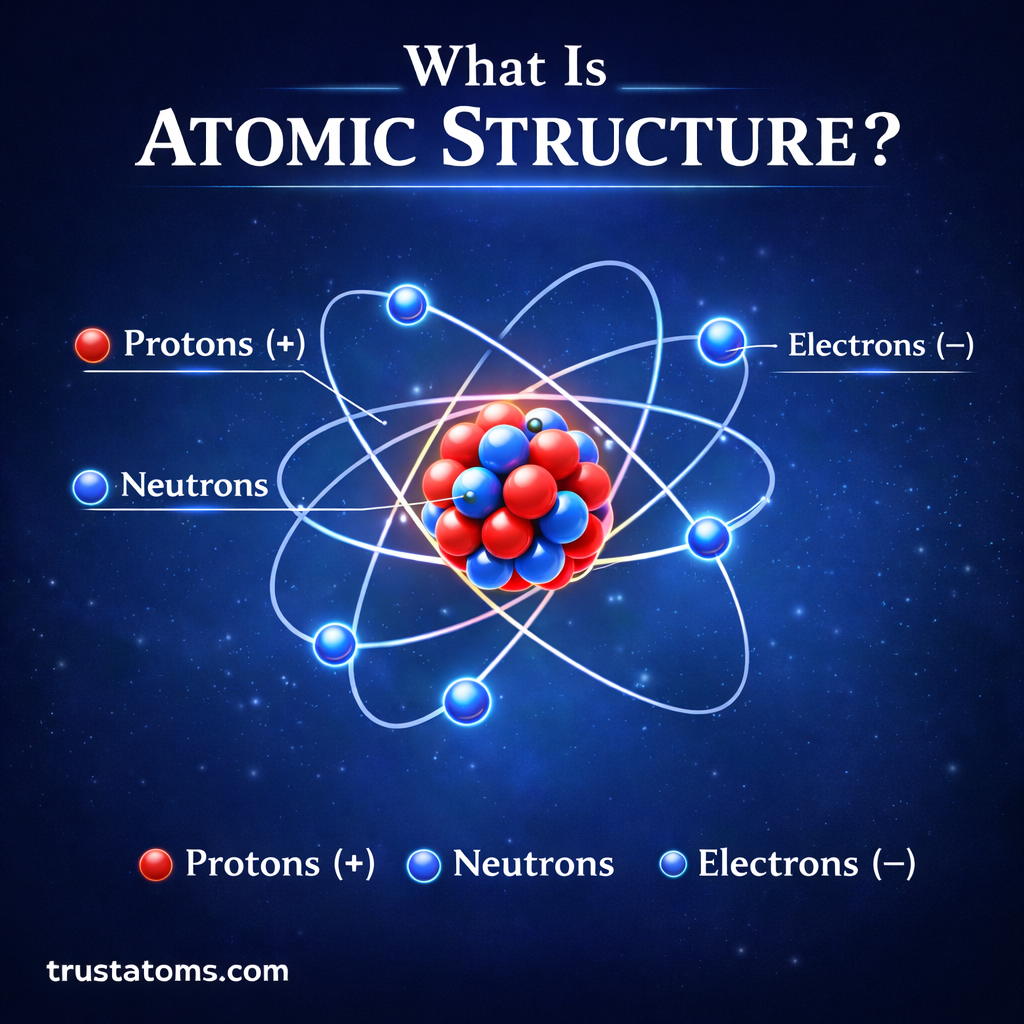
Atomic structure refers to the arrangement of particles inside an atom. It explains how protons, neutrons, and electrons are organized and how they determine the properties of elements.
Everything around you — air, water, metals, living organisms — is made of atoms. Understanding atomic structure is key to understanding chemistry, electricity, radiation, and even modern technology.
Atomic structure forms one of the foundations of physics.
What Is an Atom?
An atom is the smallest unit of matter that retains the properties of an element.
Each atom has two main regions:
- The nucleus (center)
- The electron cloud (surrounding region)
Although atoms are incredibly small, their internal structure determines how matter behaves.
The Nucleus
The nucleus sits at the center of the atom and contains:
- Protons (positively charged particles)
- Neutrons (neutral particles)
Protons
- Carry a positive electric charge
- Determine the element’s identity
- Equal the atomic number
For example, an atom with 6 protons is always carbon.
Neutrons
- Have no electric charge
- Add mass to the atom
- Help stabilize the nucleus
Different numbers of neutrons create isotopes of the same element.
The Electron Cloud
Electrons are negatively charged particles that move around the nucleus.
They do not orbit like planets in neat circles. Instead, they exist in regions called orbitals, which represent areas where electrons are most likely to be found.
Electrons:
- Determine chemical behavior
- Participate in bonding
- Control electrical properties
How Are Electrons Arranged?
Electrons occupy energy levels around the nucleus.
These levels:
- Increase in energy as they move farther from the nucleus
- Can only hold a certain number of electrons
- Follow specific quantum rules
The outermost energy level is called the valence shell.
Valence electrons play a major role in chemical reactions.
Atomic Number and Mass Number
Two important numbers describe atomic structure.
Atomic Number
- Equals the number of protons
- Identifies the element
Mass Number
- Equals protons + neutrons
- Represents the total mass of the nucleus
For example:
- Carbon has 6 protons
- Carbon-12 has 6 protons and 6 neutrons
Isotopes Explained
Isotopes are atoms of the same element with different numbers of neutrons.
They:
- Have the same number of protons
- Have different masses
- May have different stability
Some isotopes are stable, while others are radioactive.
Why Atomic Structure Matters
Atomic structure determines:
- Chemical reactions
- Electrical conductivity
- Magnetism
- States of matter
- Nuclear energy
Without understanding atomic structure, modern chemistry and physics would not exist.
The Evolution of Atomic Models
Our understanding of atomic structure developed over time.
Early Ideas
Ancient philosophers suggested matter was made of tiny indivisible particles.
Classical Atomic Model
Scientists proposed atoms as solid spheres.
Nuclear Model
Experiments revealed a dense nucleus at the center.
Quantum Model
Modern physics describes electrons as probability clouds rather than fixed orbits.
Today’s quantum mechanical model provides the most accurate description of atomic structure.
Atomic Structure and Electricity
Atomic structure explains electricity.
- Conductors allow electrons to move easily.
- Insulators hold electrons tightly.
- Semiconductors fall between the two.
This understanding led to:
- Transistors
- Microchips
- Modern electronics
Atomic Structure and Chemical Bonding

Atoms combine to form molecules through chemical bonds.
Bonds form when atoms:
- Share electrons
- Transfer electrons
- Attract through electrostatic forces
The structure of the outer electron shell determines how atoms bond.
Common Misconceptions About Atomic Structure
Some misunderstandings include:
- Atoms are mostly empty space.
- Electrons do not orbit like tiny planets.
- The nucleus is extremely small compared to the overall atom.
Although atoms are tiny, the forces inside them are incredibly strong.
Why Atomic Structure Is Foundational in Physics
Atomic structure connects multiple fields of science:
- Chemistry
- Nuclear physics
- Solid-state physics
- Quantum mechanics
- Astrophysics
It explains how stars produce energy, how radiation works, and how materials behave.
Understanding atomic structure unlocks a deeper understanding of matter itself.
Final Thoughts
Atomic structure describes how protons, neutrons, and electrons are arranged inside an atom.
The nucleus contains protons and neutrons, while electrons occupy energy levels around it. This arrangement determines the identity and behavior of elements.
From chemical reactions to modern electronics, atomic structure shapes the physical world.
By studying atomic structure, scientists uncover the building blocks of everything in the universe.







