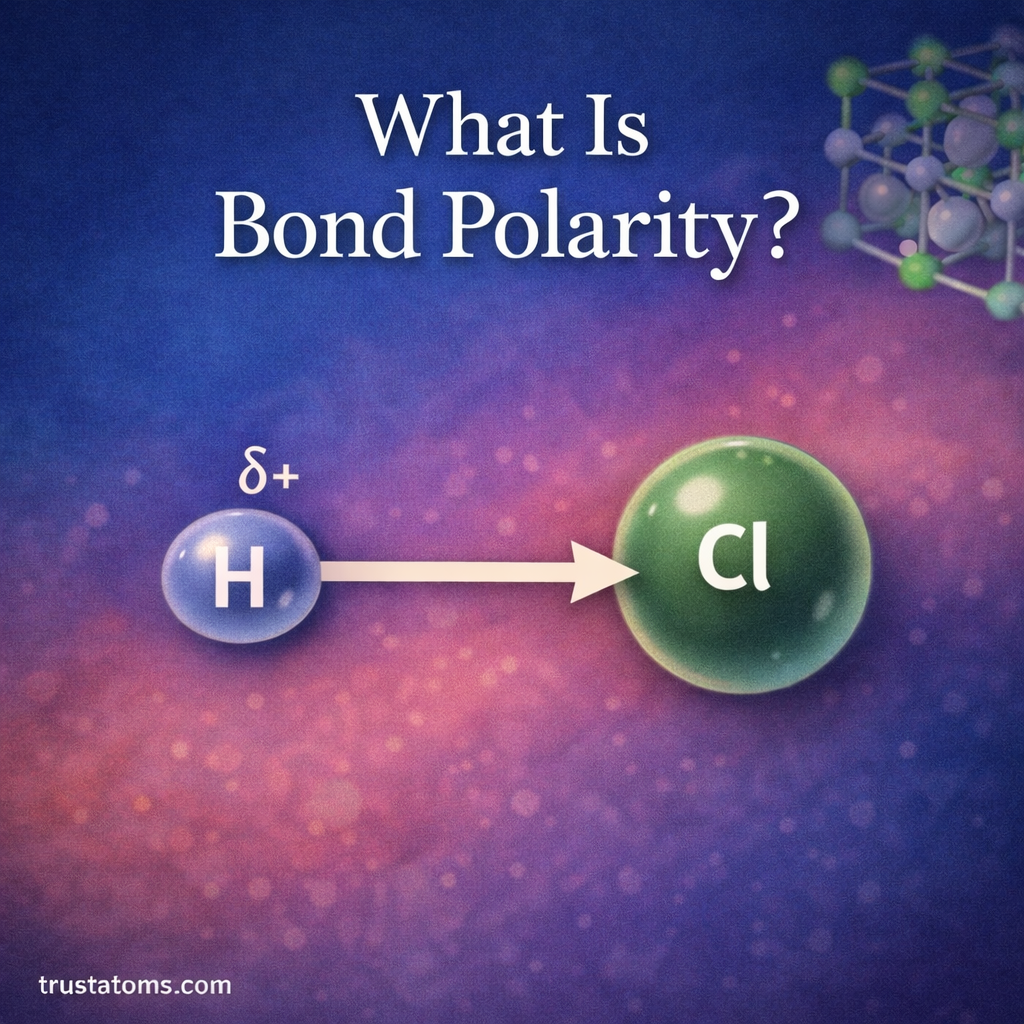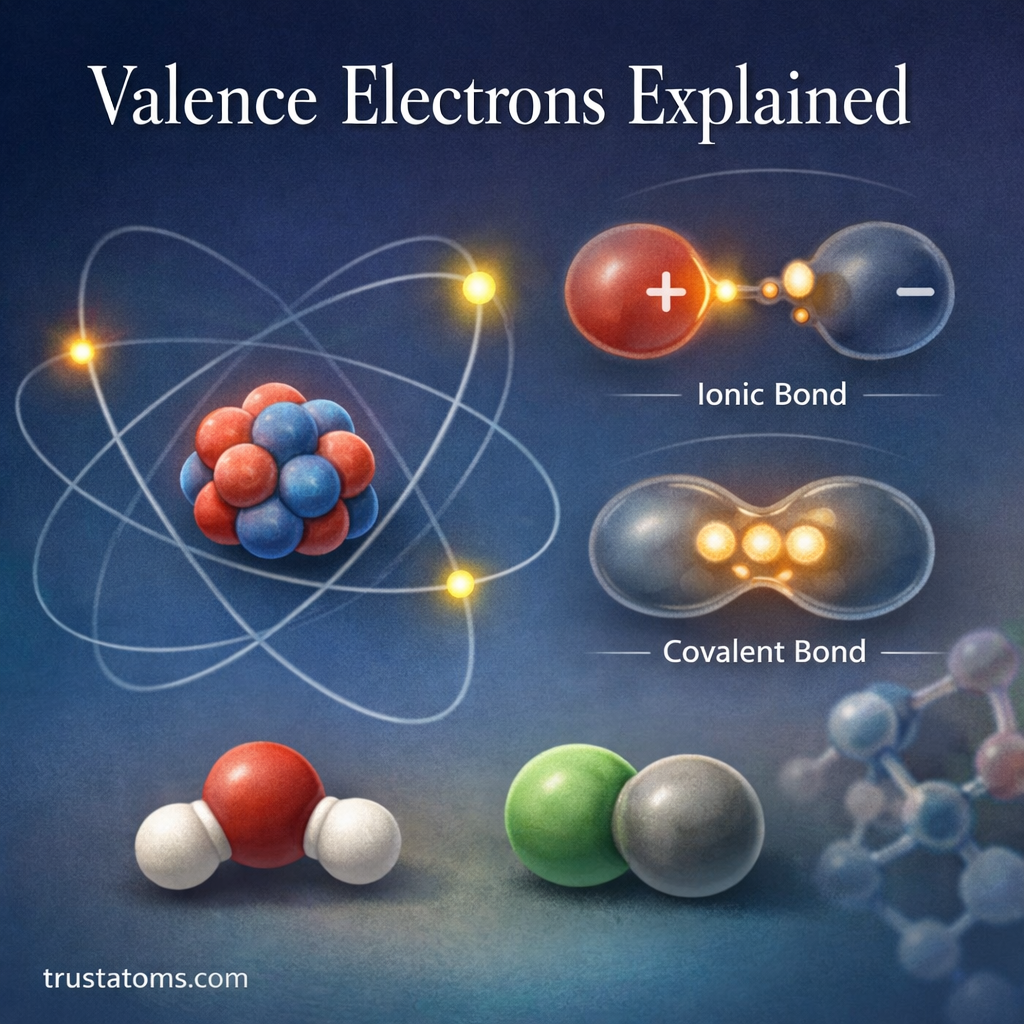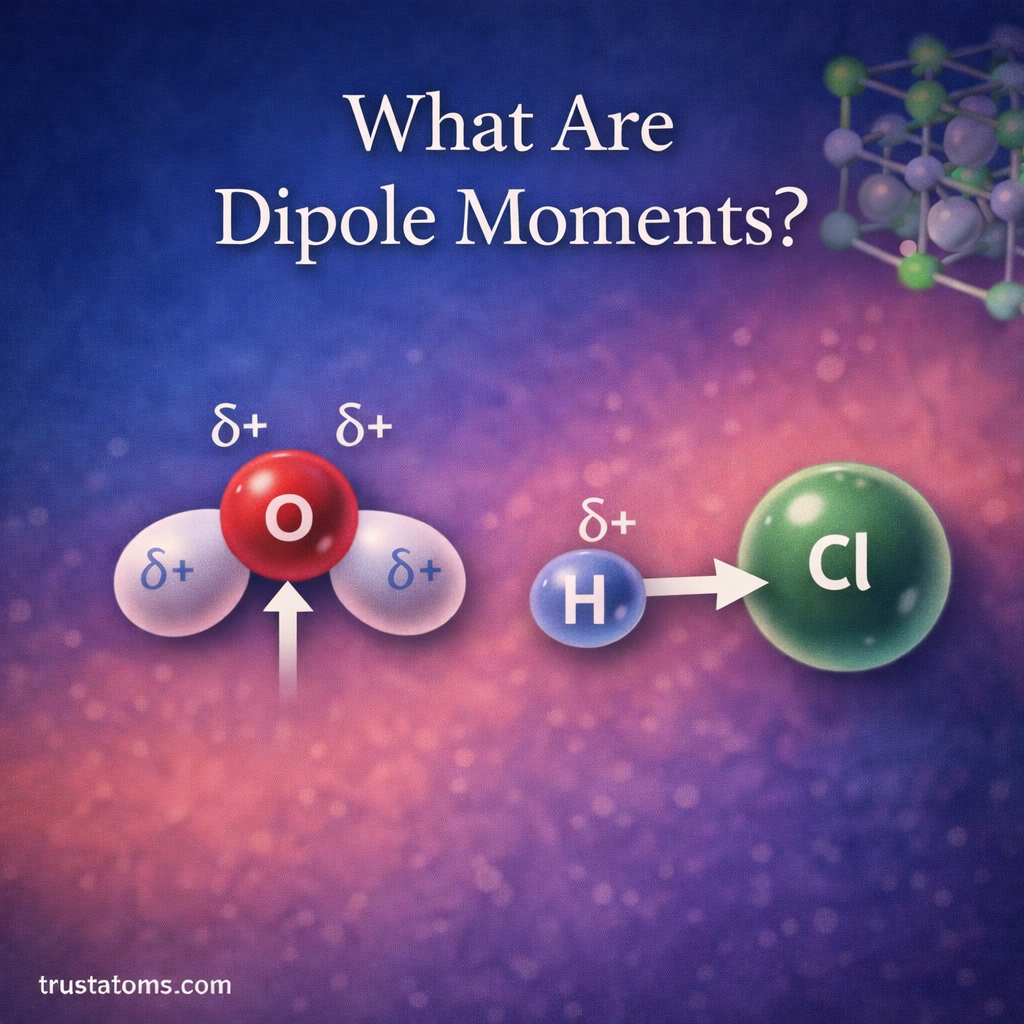
Dipole moments are an important concept in chemistry that describe how electrical charge is distributed within a molecule. When electrons are not shared evenly between atoms, parts of the molecule become slightly positive or slightly negative.
This separation of charge creates a dipole, which affects how molecules interact with each other. Dipole moments help chemists understand molecular polarity, intermolecular forces, solubility, and chemical reactions.
By studying dipole moments, scientists can predict how molecules behave in liquids, gases, biological systems, and chemical reactions.
Continue reading “What Are Dipole Moments?”