Hybridization is a concept in chemistry that explains how atoms form chemical bonds by mixing their atomic orbitals. It helps scientists understand molecular shapes, bonding patterns, and how atoms share electrons to create stable molecules.
Without hybridization, many molecules would appear to break the basic rules of orbital theory. By combining orbitals into new hybrid orbitals, atoms can form bonds that match the shapes observed in real molecules.
Hybridization is especially important in organic chemistry, where carbon atoms frequently form multiple bonds and complex structures.
Understanding Hybridization in Simple Terms
Hybridization occurs when atomic orbitals from the same atom combine to form new orbitals called hybrid orbitals.
These hybrid orbitals:
- Have different shapes and energies than the original orbitals
- Are arranged to minimize electron repulsion
- Allow atoms to form stronger and more stable chemical bonds
The most common orbitals involved in hybridization are:
- s orbitals
- p orbitals
- Sometimes d orbitals (in heavier elements)
By mixing these orbitals, atoms can create bonding arrangements that better explain the geometry of molecules.
Why Hybridization Matters
Hybridization helps chemists understand several important aspects of molecular structure.
It explains:
- The shape of molecules
- How atoms arrange themselves in space
- Why certain molecules form specific bond angles
- The stability of chemical bonds
For example, without hybridization, carbon would only be able to form two bonds. Hybridization allows carbon to form four strong bonds, which explains the wide variety of organic molecules found in nature.
Types of Hybridization
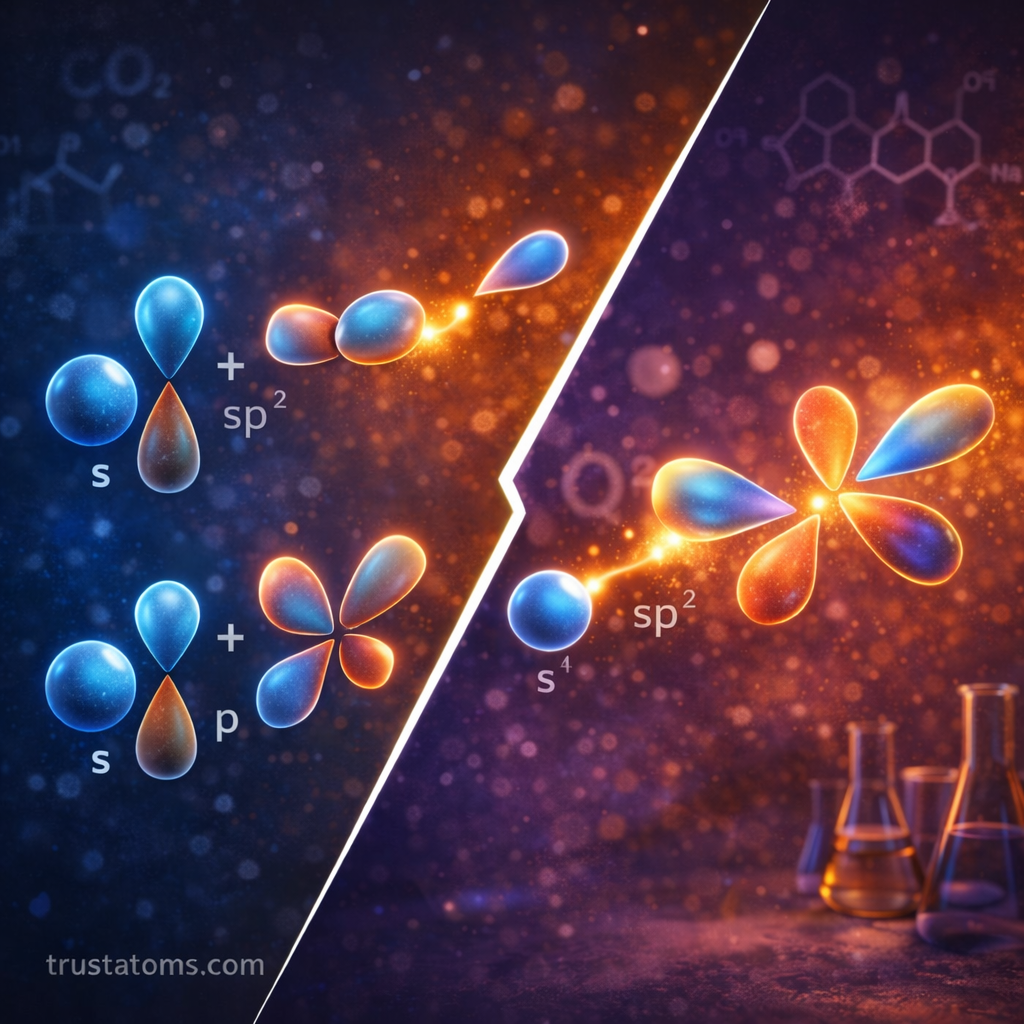
There are several common types of hybridization based on how orbitals combine.
sp Hybridization
In sp hybridization:
- One s orbital
- One p orbital
combine to form two sp hybrid orbitals.
Key characteristics:
- Linear molecular shape
- Bond angle of 180°
Example molecules:
- Acetylene (C₂H₂)
- Carbon dioxide (CO₂)
sp² Hybridization
In sp² hybridization:
- One s orbital
- Two p orbitals
combine to form three sp² hybrid orbitals.
Key characteristics:
- Trigonal planar shape
- Bond angle of 120°
Example molecules:
- Ethylene (C₂H₄)
- Boron trifluoride (BF₃)
sp³ Hybridization
In sp³ hybridization:
- One s orbital
- Three p orbitals
combine to form four sp³ hybrid orbitals.
Key characteristics:
- Tetrahedral shape
- Bond angle of 109.5°
Example molecules:
- Methane (CH₄)
- Water (H₂O)
- Ammonia (NH₃)
sp³d Hybridization
This type involves:
- One s orbital
- Three p orbitals
- One d orbital
These combine to form five hybrid orbitals.
Key characteristics:
- Trigonal bipyramidal shape
- Bond angles of 90° and 120°
Example molecules:
- Phosphorus pentachloride (PCl₅)
sp³d² Hybridization
In this hybridization:
- One s orbital
- Three p orbitals
- Two d orbitals
combine to form six hybrid orbitals.
Key characteristics:
- Octahedral molecular shape
- Bond angle of 90°
Example molecules:
- Sulfur hexafluoride (SF₆)
How Hybridization Determines Molecular Shape
Hybridization works closely with electron pair repulsion principles. Electrons naturally arrange themselves to stay as far apart as possible.
Hybrid orbitals orient themselves in space to reduce repulsion, which determines the molecule’s geometry.
Common molecular shapes include:
- Linear
- Trigonal planar
- Tetrahedral
- Trigonal bipyramidal
- Octahedral
Understanding hybridization helps predict these shapes accurately.
Steps to Identify Hybridization
Chemists often follow a simple process to determine the hybridization of an atom in a molecule.
- Draw the Lewis structure of the molecule.
- Count the number of regions of electron density around the atom.
- Match the number of regions to a hybridization type.
Quick reference:
- 2 regions → sp
- 3 regions → sp²
- 4 regions → sp³
- 5 regions → sp³d
- 6 regions → sp³d²
This approach helps identify the correct orbital arrangement.
Hybridization in Organic Chemistry
Hybridization is particularly important in carbon-based molecules.
Carbon atoms frequently change hybridization depending on the bonds they form.
Examples include:
- sp³ carbon in alkanes
- sp² carbon in alkenes
- sp carbon in alkynes
These differences influence:
- Molecular shape
- Bond strength
- Chemical reactivity
Because carbon can form long chains and rings using different hybridizations, it creates the enormous diversity of organic compounds found in biology and industry.
Real-World Importance of Hybridization
Hybridization helps scientists understand the structure and behavior of molecules used in everyday life.
Applications include:
- Pharmaceutical drug design
- Materials science
- Polymer chemistry
- Environmental chemistry
- Biological molecule analysis
By understanding how orbitals combine and bonds form, chemists can design molecules with specific properties.
Final Thoughts
Hybridization is a key concept that explains how atoms combine their orbitals to form stable chemical bonds. By mixing s, p, and sometimes d orbitals, atoms create hybrid orbitals that determine the shape and bonding structure of molecules.
This concept helps chemists predict molecular geometry, understand chemical reactions, and explain the structures of many compounds found in nature and technology.
Understanding hybridization provides a deeper view of how atoms interact and how the molecular world is organized.