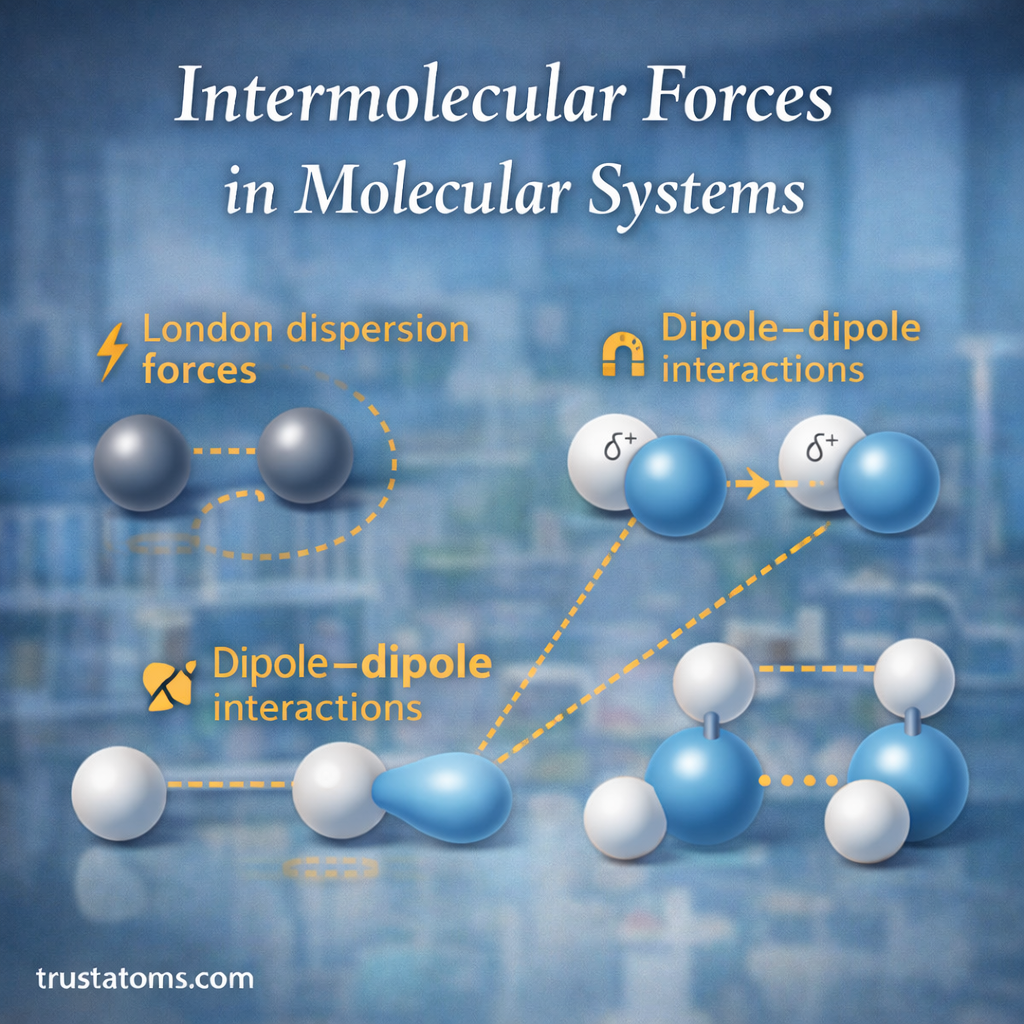
In chemistry, concentration refers to the amount of a substance (called a solute) that is present in a given amount of another substance (usually a solvent or solution). It describes how strong or diluted a chemical solution is.
For example, when sugar dissolves in water, the sugar is the solute and the water is the solvent. The concentration tells us how much sugar is dissolved in the water.
Understanding concentration is important in chemistry because it affects how reactions occur, how substances behave in solutions, and how chemical mixtures are measured and controlled in laboratories, medicine, and industry.
Understanding Solutions in Chemistry
To understand concentration, it helps to first understand what a solution is.
A solution is a homogeneous mixture composed of:
- Solute – the substance being dissolved
- Solvent – the substance doing the dissolving
Examples of common solutions include:
- Salt dissolved in water
- Sugar dissolved in tea
- Carbon dioxide dissolved in soda
The concentration describes how much solute exists relative to the solvent or total solution.
Why Concentration Matters
Concentration plays a major role in many chemical processes. Different concentrations can change how substances react, taste, behave, or affect biological systems.
Chemists measure concentration to:
- Control chemical reactions
- Prepare laboratory solutions
- Produce medicines safely
- Monitor environmental chemicals
- Analyze water and air quality
In many reactions, changing the concentration of reactants can increase or decrease reaction speed.
Common Ways to Express Concentration
Chemists use several methods to express concentration depending on the situation.
1. Molarity
Molarity is one of the most common ways to measure concentration in chemistry.
It describes the number of moles of solute dissolved in one liter of solution.
Molarity is often used in laboratory experiments because it provides precise measurements for chemical reactions.
2. Percent Concentration
Percent concentration expresses the amount of solute as a percentage of the total solution.
It is commonly used in everyday products such as:
- Cleaning solutions
- Medical disinfectants
- Food and beverages
For example, a solution labeled as 5% salt means that 5% of the mixture is salt and the rest is solvent.
3. Parts Per Million (PPM)
Parts per million is used to measure very small concentrations.
This measurement is common in environmental science for substances like:
- Air pollutants
- Chemicals in drinking water
- Trace metals in soil
Because these substances occur in extremely small amounts, ppm allows scientists to detect and measure them accurately.
Concentrated vs Dilute Solutions

Solutions are often described using general terms such as concentrated or dilute.
Concentrated Solution
A concentrated solution contains a large amount of solute relative to the solvent.
Examples include:
- Strong coffee
- Highly salted water
- Concentrated cleaning products
Dilute Solution
A dilute solution contains a smaller amount of solute relative to the solvent.
Examples include:
- Lightly salted water
- Diluted fruit juice
- Weak chemical mixtures
These terms describe relative strength but do not provide precise measurements like molarity or percentages.
Dilution in Chemistry
Dilution is the process of reducing concentration by adding more solvent.
For example:
- A concentrated acid solution is prepared in a laboratory.
- Water is added to the solution.
- The overall concentration becomes lower.
Dilution is commonly used in:
- Laboratory experiments
- Medical testing
- Pharmaceutical preparation
- Industrial chemical production
Careful dilution ensures that chemical solutions remain safe and accurate.
Concentration and Chemical Reactions
Concentration directly affects the speed of many chemical reactions.
When the concentration of reactants increases:
- More particles are present in the solution
- Collisions between particles occur more frequently
- Reaction rates often increase
When concentration decreases, reactions may occur more slowly because fewer particle collisions happen.
This concept is closely connected to chemical kinetics, the study of reaction rates.
Real-World Applications of Concentration
Concentration is used in many real-world scientific and industrial applications.
Medicine and Healthcare
Doctors and pharmacists must carefully control drug concentration to ensure treatments are safe and effective.
Examples include:
- Intravenous (IV) solutions
- Liquid medications
- Blood chemistry tests
Environmental Monitoring
Scientists measure concentration to monitor environmental health.
Examples include:
- Measuring pollutants in air
- Checking chemical levels in drinking water
- Tracking ocean salinity
Food and Beverage Industry
Concentration affects taste, preservation, and product consistency.
Examples include:
- Sugar concentration in drinks
- Salt levels in food
- Flavor intensity in sauces
Careful concentration control ensures consistent quality.
Final Thoughts
Concentration is a fundamental concept in chemistry that describes how much of a substance is present in a solution. By measuring concentration, scientists can control chemical reactions, produce medicines, monitor environmental conditions, and manufacture products safely.
Whether in laboratories, industry, or everyday life, understanding concentration helps explain how chemical mixtures behave and how solutions can be adjusted to achieve the desired results.