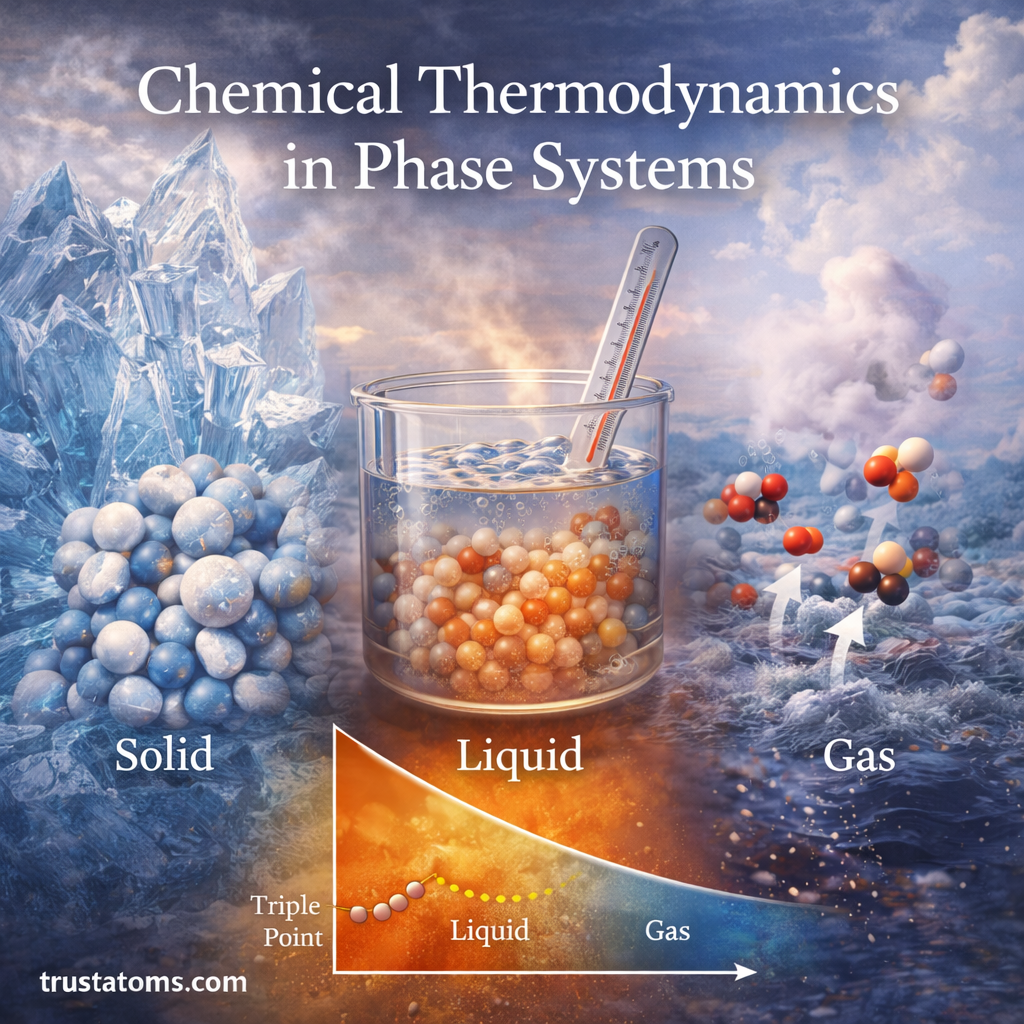
Organic chemistry is a major branch of chemistry that focuses on the study of carbon-containing compounds. These compounds form the basis of life and appear in countless materials used in everyday life, from medicines and plastics to fuels and food.
At its core, organic chemistry examines how carbon atoms bond with other elements to form molecules with unique structures and properties. Because carbon can form complex chains and rings, it is capable of creating an enormous variety of chemical compounds.
Today, organic chemistry plays a vital role in science, medicine, technology, and industry.
Understanding Organic Chemistry in Simple Terms
Organic chemistry is the study of molecules built primarily from carbon atoms. Carbon is special because it can form four chemical bonds, allowing it to create stable and complex molecular structures.
These structures may include:
- Straight chains of carbon atoms
- Branched chains
- Ring-shaped molecules
- Large biological molecules
Many important substances studied in organic chemistry include:
- Proteins
- Carbohydrates
- Fats
- DNA
- Pharmaceuticals
- Plastics
Because living organisms are made largely from carbon-based molecules, organic chemistry is closely connected to biology and biochemistry.
Why Carbon Is Central to Organic Chemistry
Carbon is unique among elements because of its bonding flexibility.
A carbon atom can:
- Form four strong covalent bonds
- Bond with other carbon atoms
- Bond with elements such as hydrogen, oxygen, nitrogen, sulfur, and halogens
- Create long chains and complex structures
This ability allows carbon to produce millions of different compounds.
For example, carbon structures can form:
- Simple molecules like methane
- Long hydrocarbon chains found in fuels
- Complex molecules such as proteins and enzymes
Because of this versatility, carbon chemistry became its own field of study.
Types of Organic Compounds
Organic compounds come in many forms, each with different structures and chemical behaviors.
Some common categories include:
Hydrocarbons
Hydrocarbons contain only carbon and hydrogen atoms.
Examples include:
- Methane
- Ethane
- Propane
- Gasoline components
These compounds are widely used as energy sources and fuels.
Alcohols
Alcohols contain a carbon atom bonded to an oxygen and hydrogen group.
Examples include:
- Ethanol (found in beverages and fuel)
- Isopropyl alcohol (used as a disinfectant)
Alcohols are common in both industrial chemistry and biology.
Carboxylic Acids
Carboxylic acids contain a carbon atom bonded to a carboxyl group, which gives them acidic properties.
Examples include:
- Acetic acid (vinegar)
- Fatty acids in biological systems
These compounds are important in food chemistry and biological metabolism.
Polymers
Polymers are large molecules made from repeating units of smaller molecules.
Examples include:
- Plastics
- Synthetic fibers
- Rubber
- Many biological molecules
Organic chemistry plays a major role in polymer science and materials engineering.
Organic Chemistry and Living Systems
Organic chemistry is closely connected to the chemistry of life.
Many biological molecules are organic compounds, including:
- Proteins that control cellular processes
- Carbohydrates that store and supply energy
- Lipids that form cell membranes
- DNA that carries genetic information
Biochemistry, a related field, studies how these organic molecules function within living organisms.
Understanding these molecules helps scientists study health, disease, and biological processes.
Applications of Organic Chemistry

Organic chemistry has a wide range of real-world applications that affect everyday life.
Medicine and Pharmaceuticals
Many drugs are organic molecules designed to interact with biological systems.
Examples include:
- Antibiotics
- Pain relievers
- Vaccines
- Hormone therapies
Organic chemistry allows scientists to design and synthesize new medications.
Materials and Plastics
Modern materials often rely on organic compounds.
Examples include:
- Plastic containers
- Synthetic fabrics
- Adhesives
- Insulation materials
Organic chemistry enables the development of new materials with specific properties.
Energy and Fuels
Many energy sources are based on organic molecules.
Examples include:
- Petroleum
- Natural gas
- Biofuels
Understanding these compounds helps improve fuel efficiency and develop renewable energy alternatives.
Agriculture and Food
Organic chemistry is also used in food science and agriculture.
Applications include:
- Food preservation
- Flavor compounds
- Fertilizers
- Crop protection chemicals
These advances help support global food production.
How Organic Chemistry Is Studied
Organic chemists study molecules by examining their structure, reactions, and properties.
Important areas of study include:
- Molecular structure
- Chemical reactions
- Reaction mechanisms
- Synthesis of new compounds
- Analytical techniques for identifying molecules
Laboratory techniques often involve spectroscopy, chromatography, and chemical synthesis.
Through these methods, scientists can determine how molecules behave and how they can be modified.
Why Organic Chemistry Matters
Organic chemistry is one of the most important fields in modern science because it helps explain how complex molecules form and interact.
The knowledge gained from organic chemistry contributes to:
- Medical breakthroughs
- New materials
- Sustainable energy technologies
- Advances in biotechnology
- Improved food production
By understanding how carbon-based molecules work, scientists can design solutions to many scientific and technological challenges.
Final Thoughts
Organic chemistry is the branch of chemistry that studies carbon-containing compounds and their reactions. Because carbon can form complex and diverse molecules, organic chemistry covers an enormous range of substances found in nature and industry.
From the chemistry of living organisms to the development of medicines, fuels, and materials, organic chemistry plays a crucial role in understanding and improving the world around us.