Acid–base reactions are among the most fundamental chemical reactions in nature. They occur when an acid interacts with a base, typically resulting in the transfer of a proton (hydrogen ion) from one substance to another.
These reactions are responsible for many everyday processes—from digestion in the human body to the effectiveness of cleaning products and even the chemistry of soil that supports plant growth.
Understanding acid–base reactions helps explain how substances neutralize each other and how chemical balance is maintained in both natural and industrial systems.
The Basic Idea of an Acid–Base Reaction
An acid–base reaction occurs when an acid donates a hydrogen ion (H⁺) and a base accepts that hydrogen ion.
In simple terms:
- Acids give away protons.
- Bases receive protons.
This transfer of protons is the key feature that defines an acid–base reaction in chemistry.
When the reaction occurs, new substances are formed—often including water and a salt.
Definitions of Acids and Bases
Chemists use several models to describe acids and bases. The two most commonly taught are the Arrhenius definition and the Brønsted–Lowry definition.
Arrhenius Definition
According to the Arrhenius model:
- Acids produce hydrogen ions (H⁺) when dissolved in water.
- Bases produce hydroxide ions (OH⁻) when dissolved in water.
Example:
- Hydrochloric acid releases hydrogen ions in water.
- Sodium hydroxide releases hydroxide ions.
These ions interact during an acid–base reaction.
Brønsted–Lowry Definition
The Brønsted–Lowry theory expands the concept of acids and bases.
- Acid: a proton donor
- Base: a proton acceptor
This definition works even in reactions that do not involve water, making it more widely applicable.
For example:
- Ammonia acts as a base by accepting a proton.
- Acetic acid acts as an acid by donating a proton.
What Happens During the Reaction?
During an acid–base reaction, a proton transfers from the acid to the base.
This creates two new substances called a conjugate acid–base pair.
The process typically follows these steps:
- The acid releases a hydrogen ion.
- The base accepts the hydrogen ion.
- New molecules form from the rearranged components.
The result is a chemical change that alters the properties of the substances involved.
Neutralization Reactions
One of the most common types of acid–base reactions is neutralization.
Neutralization occurs when an acid and a base react to produce:
- Water
- A salt
Example concept:
- An acid reacts with a base.
- Hydrogen ions combine with hydroxide ions.
- Water forms, and the remaining ions form a salt.
This process reduces the acidic or basic properties of the solution.
Examples of Acid–Base Reactions in Everyday Life
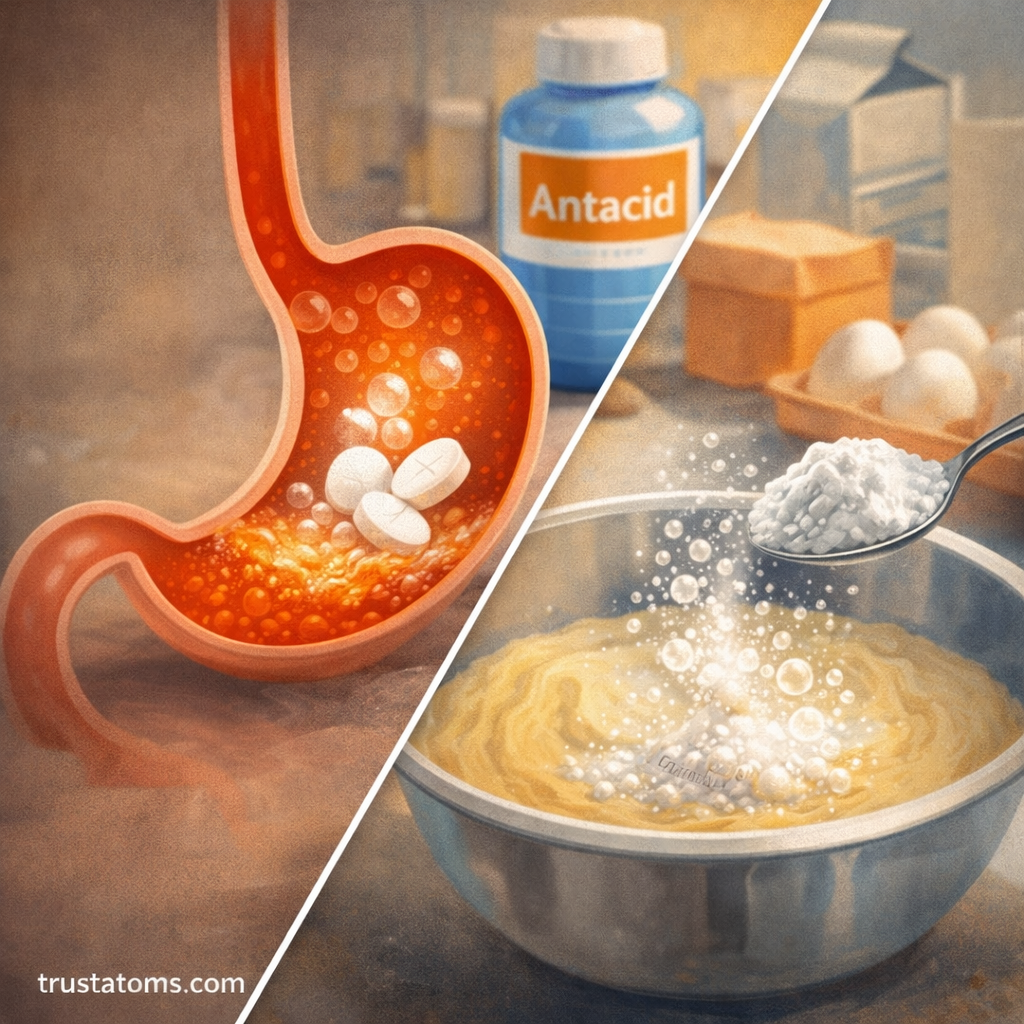
Acid–base chemistry appears in many ordinary situations.
1. Antacids and Digestion
Stomach acid contains hydrochloric acid.
Antacid tablets contain basic compounds that neutralize this acid and reduce discomfort from heartburn.
2. Baking
Baking soda reacts with acidic ingredients in recipes.
The reaction produces carbon dioxide gas, which helps baked goods rise.
3. Soil Chemistry
Farmers often add lime (a basic compound) to acidic soil.
This neutralization improves soil conditions for plant growth.
4. Cleaning Products
Many household cleaners rely on basic substances to break down grease and acidic residues.
Strong vs. Weak Acids and Bases
Not all acids and bases behave the same way in reactions.
Strong Acids and Bases
Strong acids and bases fully dissociate in water, meaning they release all of their available ions.
Examples include:
- Strong acids such as hydrochloric acid
- Strong bases such as sodium hydroxide
These substances react quickly and completely.
Weak Acids and Bases
Weak acids and bases only partially dissociate in water.
Examples include:
- Acetic acid (found in vinegar)
- Ammonia
Because they release fewer ions, their reactions are generally less intense.
The Role of pH
Acid–base reactions are closely connected to the pH scale, which measures how acidic or basic a solution is.
The pH scale typically ranges from:
- 0–6: Acidic
- 7: Neutral
- 8–14: Basic (alkaline)
During neutralization, the pH of a solution moves closer to neutral as acids and bases react.
Maintaining the correct pH is essential in systems such as:
- Human blood chemistry
- Industrial chemical processes
- Environmental water systems
Why Acid–Base Reactions Matter
Acid–base reactions are essential to chemistry because they control how substances interact in many environments.
They play important roles in:
- Biological processes
- Food chemistry
- Environmental science
- Medicine
- Industrial manufacturing
By understanding how acids donate protons and bases accept them, scientists can predict chemical behavior and design processes that depend on precise chemical balance.
Final Thoughts
Acid–base reactions are a central concept in chemistry, describing how substances exchange hydrogen ions and transform into new compounds. From neutralizing stomach acid to supporting plant growth in soil, these reactions occur constantly in both natural and human-made systems.
Learning how acids and bases interact provides a foundation for understanding many other chemical processes, making acid–base chemistry one of the most important topics in the study of science.