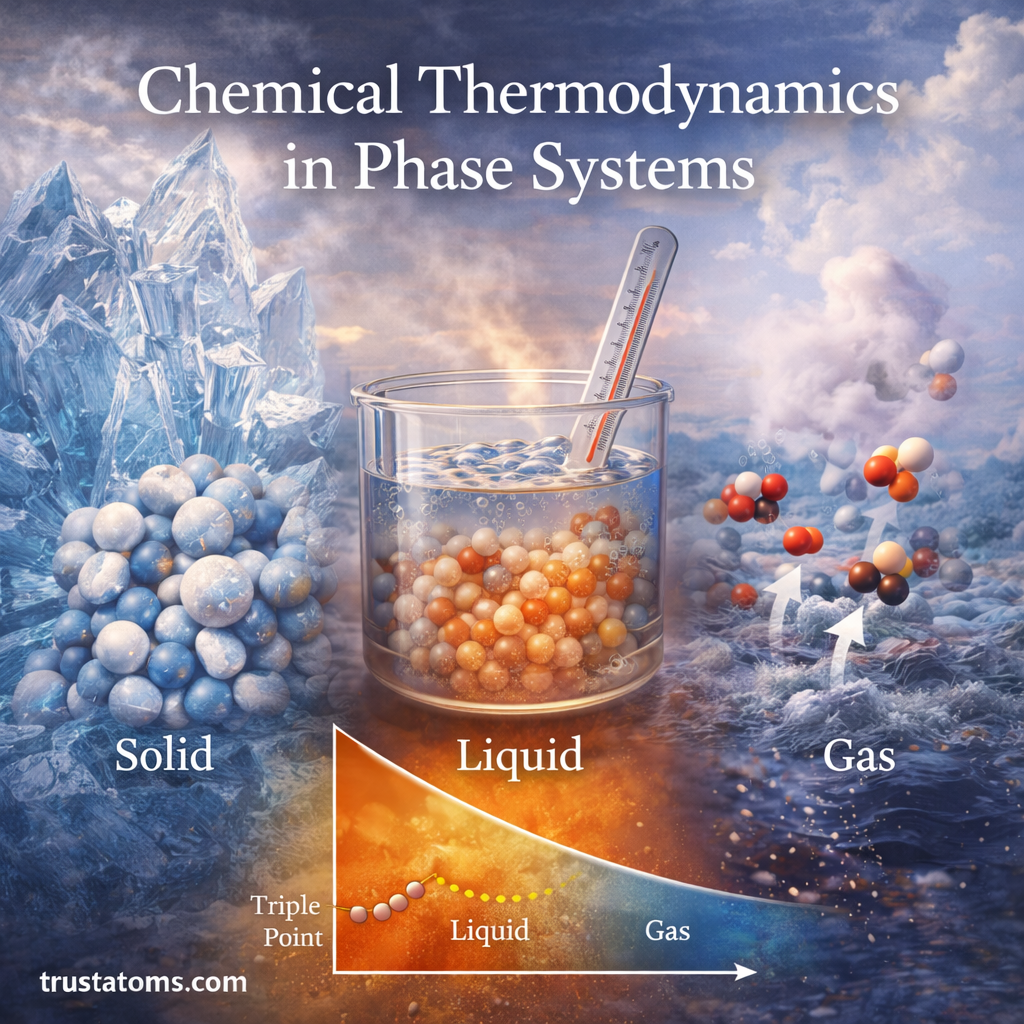
Chemical thermodynamics helps scientists understand how energy, temperature, and pressure influence chemical systems. One of its most important applications involves phase systems, where substances exist in different physical states such as solid, liquid, or gas.
In many chemical and industrial processes, materials move between phases or exist in equilibrium between multiple phases. Chemical thermodynamics provides the tools needed to predict how these systems behave, how energy flows through them, and under what conditions phase changes occur.
Understanding thermodynamics in phase systems is essential in fields such as materials science, chemical engineering, environmental chemistry, and energy production.
What Is a Phase System?
A phase system refers to a chemical system that contains one or more phases. A phase is a physically and chemically uniform portion of a system separated from other regions by distinct boundaries.
Common phases include:
- Solids
- Liquids
- Gases
- Plasma (in specialized systems)
A system can contain:
- Single-phase systems, such as pure liquid water
- Two-phase systems, such as liquid water and water vapor
- Multi-phase systems, such as ice, liquid water, and vapor together
Each phase has its own properties, including density, structure, and molecular arrangement.
Key Thermodynamic Variables in Phase Systems
Several thermodynamic variables determine how phase systems behave. These variables influence phase stability, phase transitions, and equilibrium conditions.
Temperature
Temperature controls the kinetic energy of molecules in a system.
As temperature increases:
- Molecular motion increases
- Intermolecular forces weaken
- Phase transitions may occur
For example:
- Ice melts into liquid water when temperature rises above its melting point.
- Liquid water becomes vapor when it reaches its boiling point.
Pressure
Pressure also strongly influences phase behavior.
Higher pressure tends to:
- Compress gases
- Stabilize condensed phases (liquids and solids)
- Shift equilibrium conditions in phase systems
Pressure effects are especially important in systems involving gases or deep geological environments.
Composition
In multi-component systems, the chemical composition determines which phases can form.
For example:
- Salt dissolved in water creates a liquid solution phase
- Alloys contain mixtures of metals with different solid phases
Changes in composition can alter phase boundaries and equilibrium conditions.
Phase Equilibrium
Phase equilibrium occurs when multiple phases coexist without any net change over time. At equilibrium, molecules may still move between phases, but the rates of transfer are balanced.
Examples of phase equilibrium include:
- Water and water vapor in a sealed container
- Ice floating in liquid water at the melting point
- Liquid and vapor phases in boiling liquids
At equilibrium:
- Temperature remains constant
- Pressure remains constant
- Chemical potentials of components are equal between phases
Thermodynamics allows scientists to determine when equilibrium conditions occur.
Phase Transitions
A phase transition is a transformation from one physical state to another.
Common phase transitions include:
- Melting – solid to liquid
- Freezing – liquid to solid
- Vaporization – liquid to gas
- Condensation – gas to liquid
- Sublimation – solid directly to gas
- Deposition – gas directly to solid
These changes occur when energy is absorbed or released by the system.
For example:
- Melting requires energy to break intermolecular forces.
- Freezing releases energy as molecules organize into solid structures.
Thermodynamics helps quantify the energy involved in these transitions.
The Phase Rule
One of the most important principles governing phase systems is the Gibbs Phase Rule. This rule describes how many independent variables can be changed without altering the number of phases in equilibrium.
The rule relates three key factors:
- Number of phases
- Number of components
- Degrees of freedom
Degrees of freedom represent the number of variables—such as temperature, pressure, or composition—that can be adjusted independently.
The phase rule helps scientists determine:
- Stability conditions for different phases
- The number of phases that can coexist
- How changes in temperature or pressure affect the system
Phase Diagrams
Phase diagrams visually represent how phases change under different conditions.
These diagrams typically display relationships between:
- Temperature
- Pressure
- Composition
Phase diagrams allow scientists to identify regions where certain phases are stable.
Common features of phase diagrams include:
Phase Boundaries
Lines separating different phase regions indicate where phase transitions occur.
Triple Point
The triple point represents the unique condition where three phases coexist in equilibrium.
For example, water’s triple point occurs when ice, liquid water, and vapor exist simultaneously under specific temperature and pressure conditions.
Critical Point
The critical point marks the temperature and pressure above which a liquid and gas phase become indistinguishable.
Beyond this point, the substance forms a supercritical fluid with properties of both liquid and gas.
Thermodynamic Potentials in Phase Systems
Thermodynamic potentials help determine which phases are stable under specific conditions.
Important thermodynamic quantities include:
Internal Energy
Internal energy represents the total energy stored within a system.
It includes:
- Molecular kinetic energy
- Potential energy from intermolecular interactions
Enthalpy
Enthalpy measures heat content at constant pressure.
Phase transitions often involve enthalpy changes, such as:
- Heat of fusion (melting)
- Heat of vaporization
Gibbs Free Energy
Gibbs free energy is especially important for predicting phase stability.
A system naturally moves toward the state with lower Gibbs free energy.
At equilibrium:
- Gibbs free energy is minimized
- No spontaneous changes occur between phases
Thermodynamic calculations often rely on Gibbs free energy to determine which phases are favored under given conditions.
Real-World Applications of Phase Thermodynamics

Understanding chemical thermodynamics in phase systems supports many technologies and industrial processes.
Materials Science
Phase diagrams guide the development of metals, alloys, ceramics, and semiconductors.
Controlling phase transformations helps engineers design materials with specific properties such as strength, hardness, and conductivity.
Chemical Manufacturing
Industrial processes often depend on controlling phase behavior.
Examples include:
- Distillation
- Crystallization
- Gas separation
- Liquid extraction
These methods rely on differences in phase stability to separate chemical components.
Energy Systems
Phase thermodynamics plays a role in energy technologies such as:
- Refrigeration cycles
- Power plant steam systems
- Supercritical fluid extraction
- Hydrogen storage technologies
Accurate thermodynamic models allow engineers to design more efficient energy systems.
Environmental and Atmospheric Science
Phase transitions also occur naturally in environmental systems.
Examples include:
- Cloud formation
- Ice formation in the atmosphere
- Evaporation and condensation in the water cycle
Thermodynamics helps scientists understand and predict these processes.
Final Thoughts
Chemical thermodynamics provides a powerful framework for understanding how substances behave in systems containing multiple phases. By analyzing how temperature, pressure, and composition affect phase stability, scientists can predict when phase transitions occur and how systems reach equilibrium.
Concepts such as phase diagrams, thermodynamic potentials, and the phase rule allow researchers to analyze complex chemical systems and design processes that control phase behavior.
From industrial manufacturing to natural environmental processes, thermodynamics in phase systems remains a cornerstone of modern chemistry and chemical engineering.