Electron spin is a fundamental property of electrons that plays an important role in modern spectroscopic techniques. Although electrons are often described as tiny particles orbiting the nucleus, they also possess an intrinsic quantum property called spin, which behaves like a miniature magnetic moment.
In spectroscopic analysis, electron spin allows scientists to study the magnetic and electronic behavior of atoms, molecules, and materials. Techniques that detect spin interactions provide valuable information about molecular structure, bonding, and chemical environments.
Understanding electron spin is essential for interpreting data from methods such as electron spin resonance (ESR) and electron paramagnetic resonance (EPR) spectroscopy.
What Is Electron Spin?
Electron spin is an intrinsic form of angular momentum carried by electrons. Unlike classical spinning objects, electron spin is a quantum mechanical property that does not involve physical rotation.
Each electron has two possible spin states:
- Spin-up ( +½ )
- Spin-down ( −½ )
These two orientations represent how an electron behaves in a magnetic field.
Because electrons carry charge, their spin produces a small magnetic field. This magnetic property allows scientists to detect and analyze electrons using spectroscopic techniques.
Why Electron Spin Matters in Spectroscopy
Electron spin affects how atoms and molecules interact with magnetic fields and electromagnetic radiation. These interactions create measurable signals in spectroscopic instruments.
Electron spin analysis helps scientists understand:
- Electronic structure of atoms and molecules
- Chemical bonding environments
- Free radical behavior
- Transition metal complexes
- Reaction mechanisms involving unpaired electrons
Spin-based spectroscopy is particularly useful for studying paramagnetic substances, which contain unpaired electrons.
Paired vs. Unpaired Electrons
Electron spin becomes most important when electrons are unpaired.
Paired Electrons
In most molecules, electrons exist in pairs within orbitals.
Characteristics:
- Opposite spins
- Magnetic effects cancel out
- Diamagnetic behavior
- No EPR signal
Examples include water, methane, and most organic molecules.
Unpaired Electrons
When an electron occupies an orbital alone, it becomes unpaired.
Characteristics:
- Net magnetic moment
- Paramagnetic behavior
- Detectable by spin-based spectroscopy
Unpaired electrons appear in:
- Free radicals
- Transition metal ions
- Certain excited states of molecules
These species are ideal for spin spectroscopy.
Electron Spin and Magnetic Fields
When a molecule containing unpaired electrons is placed in an external magnetic field, the spin states split into different energy levels.
This phenomenon is known as Zeeman splitting.
In a magnetic field:
- Spin-up and spin-down electrons have different energies
- Electrons can transition between these states
- These transitions absorb microwave radiation
Spectroscopic instruments measure this absorption to reveal molecular details.
Electron Paramagnetic Resonance (EPR) Spectroscopy
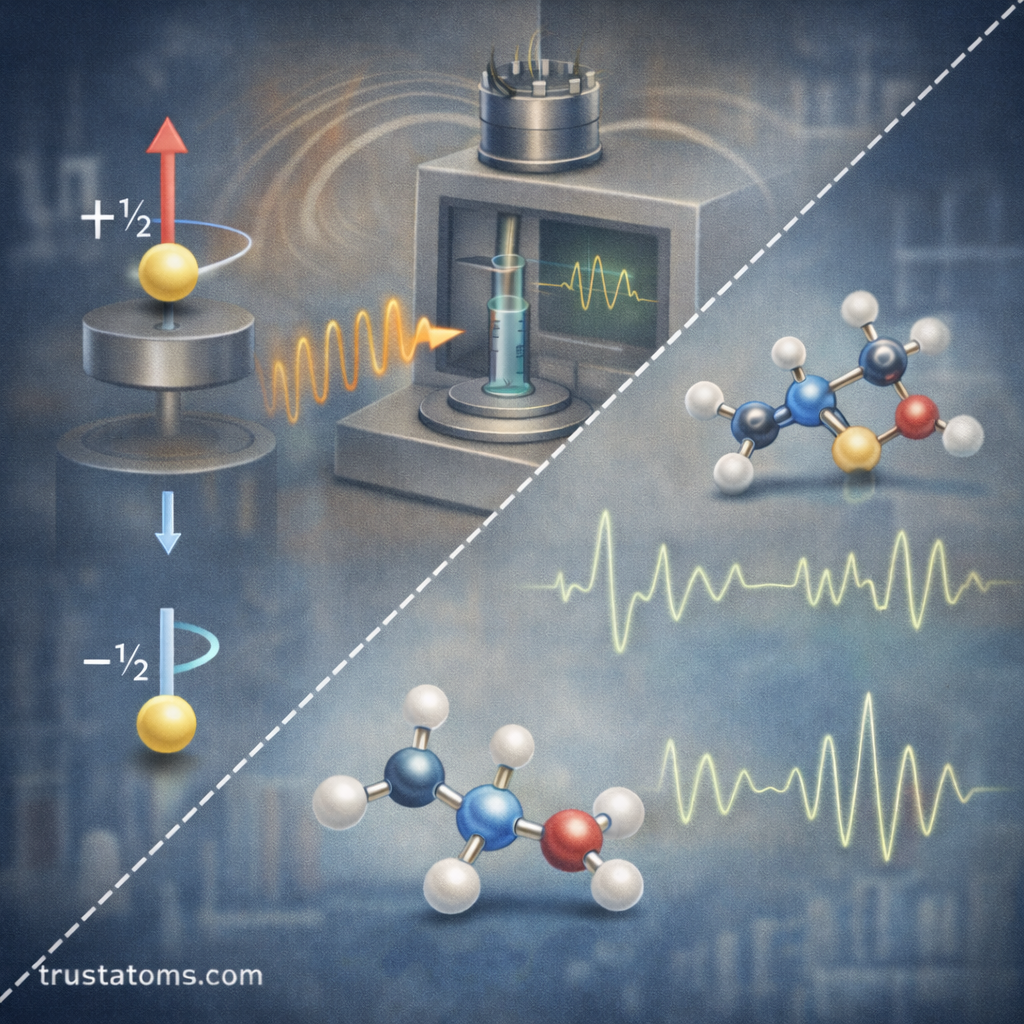
Electron Paramagnetic Resonance (EPR), also called Electron Spin Resonance (ESR), is the primary technique used to study electron spin.
EPR spectroscopy detects transitions between electron spin states in a magnetic field.
Basic Principle
The technique works through three main components:
- A strong external magnetic field
- Microwave radiation
- A sample containing unpaired electrons
When the microwave energy matches the energy difference between spin states, resonance occurs.
The instrument detects this resonance signal.
Information Obtained from EPR Spectroscopy
EPR spectroscopy provides detailed insights into the electronic structure of molecules.
Chemists use it to determine:
- Presence of free radicals
- Local chemical environment
- Identity of paramagnetic metal ions
- Molecular structure
- Electron distribution
Because electron spins are extremely sensitive to their surroundings, EPR signals often reveal subtle molecular interactions.
Hyperfine Splitting
One important feature of spin spectroscopy is hyperfine splitting.
This occurs when electron spins interact with nearby atomic nuclei.
The result is a splitting of the resonance signal into multiple smaller peaks.
Hyperfine interactions provide valuable information about:
- Nearby atoms
- Bonding structure
- Electron density distribution
For example, hydrogen atoms attached to a radical often produce distinctive hyperfine patterns in EPR spectra.
Applications of Electron Spin Spectroscopy
Electron spin spectroscopy is widely used across many scientific disciplines.
Chemistry
In chemical research, EPR helps identify:
- Reaction intermediates
- Free radical species
- Catalytic processes
- Transition metal complexes
It is especially useful in studying short-lived molecules that are difficult to observe by other methods.
Biology
Many biological molecules contain paramagnetic centers.
EPR is used to study:
- Metalloproteins
- Enzyme active sites
- Electron transport chains
- Photosynthesis mechanisms
This makes spin spectroscopy valuable in biochemistry and molecular biology.
Materials Science
Electron spin analysis helps researchers investigate:
- Semiconductors
- Magnetic materials
- Defects in crystals
- Conductive polymers
Spin behavior often determines electrical and magnetic properties of advanced materials.
Environmental and Medical Research
EPR spectroscopy is also used to detect free radicals involved in:
- Oxidative stress
- Radiation damage
- Pollution chemistry
- Aging processes
This makes the technique important in both environmental science and medical research.
Limitations of Spin-Based Spectroscopy
Although powerful, electron spin spectroscopy has certain limitations.
Requires Unpaired Electrons
Molecules without unpaired electrons cannot be analyzed using EPR spectroscopy.
This excludes many common organic compounds.
Specialized Equipment
EPR instruments require:
- High magnetic fields
- Microwave radiation sources
- Sensitive detectors
These instruments are less common than standard spectroscopy tools.
Complex Interpretation
Interpreting spin spectra can be challenging, especially when multiple interactions occur simultaneously.
Advanced theoretical models are often required.
Relationship to Other Spectroscopic Techniques
Electron spin spectroscopy complements other analytical methods.
Common related techniques include:
- Nuclear Magnetic Resonance (NMR)
- Infrared spectroscopy (IR)
- Ultraviolet-visible spectroscopy (UV-Vis)
- Raman spectroscopy
While these methods analyze different properties, combining them provides a more complete picture of molecular structure.
For example:
- NMR studies nuclear spin
- EPR studies electron spin
Together they reveal both nuclear and electronic environments.
Future Developments in Spin Spectroscopy
Advances in technology continue to expand the capabilities of spin-based spectroscopy.
Emerging developments include:
- High-frequency EPR instruments
- Pulsed EPR techniques
- Spin-based quantum sensors
- Applications in quantum computing
These innovations allow scientists to study increasingly complex molecular systems with greater precision.
Final Thoughts
Electron spin is a fundamental quantum property that plays a central role in modern spectroscopic analysis. By observing how electron spins interact with magnetic fields and electromagnetic radiation, scientists can gain deep insights into molecular structure, chemical reactions, and material properties.
Electron paramagnetic resonance spectroscopy has become an essential tool for studying free radicals, transition metal complexes, and biological molecules. As technology continues to evolve, spin-based methods will remain an important part of chemical research and advanced materials science.