Atoms may seem simple at first glance, but their internal structure is surprisingly organized. One of the key concepts in atomic structure is the arrangement of electron shells, which are the energy levels where electrons exist around the nucleus.
Electron shells determine how atoms interact with one another, how chemical bonds form, and why elements behave differently on the periodic table. Understanding electron shells is essential for learning how chemistry works at the atomic level.
What Are Electron Shells?
Electron shells are layers of energy that surround the nucleus of an atom. Electrons occupy these shells at different distances from the nucleus depending on their energy levels.
The closer a shell is to the nucleus, the lower its energy level. Shells farther from the nucleus contain electrons with higher energy.
Key points about electron shells:
- They surround the atomic nucleus
- Each shell holds a limited number of electrons
- Electrons fill lower-energy shells first
- Outer shells influence chemical behavior
Electron shells are sometimes referred to as energy levels in atomic models.
How Electron Shells Are Organized
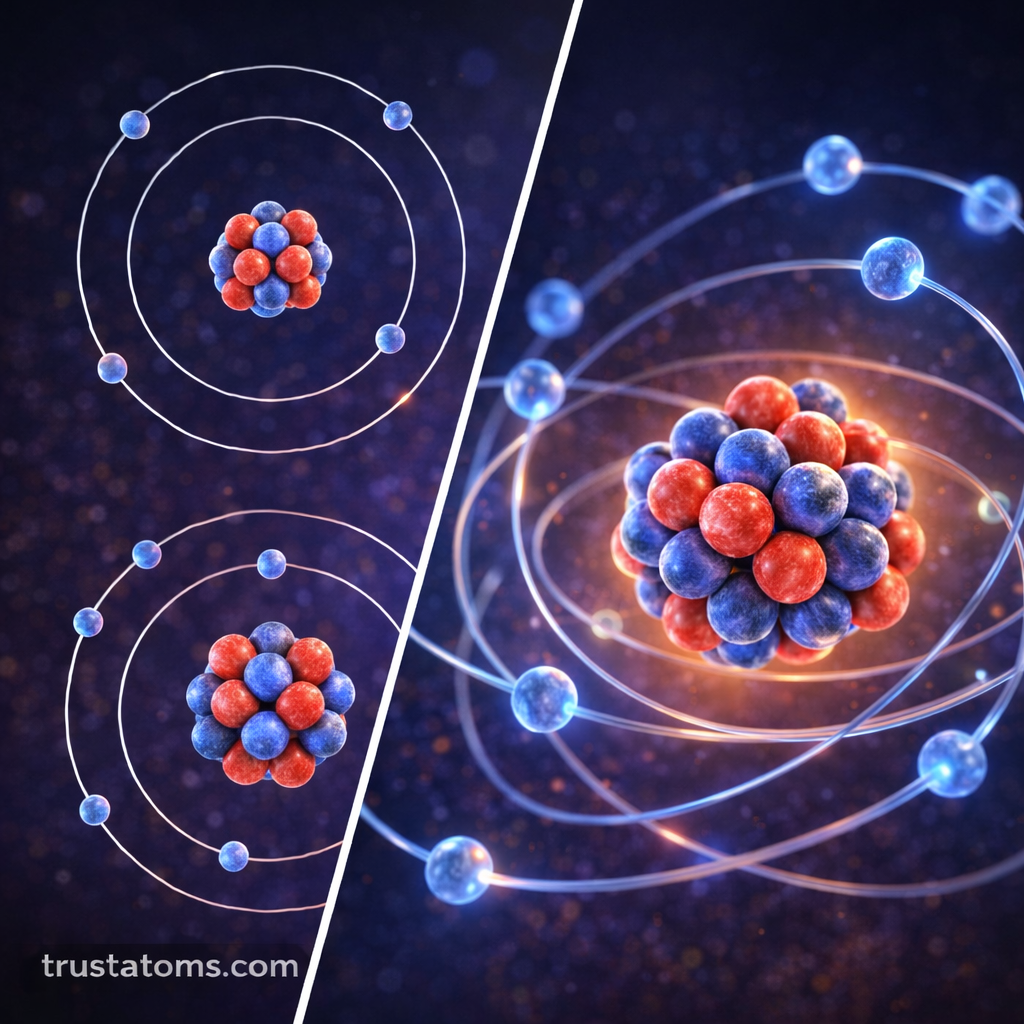
Electron shells are arranged in layers outward from the nucleus.
Scientists commonly label these shells using numbers:
- First shell (n = 1)
- Second shell (n = 2)
- Third shell (n = 3)
- Fourth shell (n = 4)
Each shell has a maximum number of electrons it can hold.
Maximum Electrons per Shell
- First shell → up to 2 electrons
- Second shell → up to 8 electrons
- Third shell → up to 18 electrons
- Fourth shell → up to 32 electrons
Electrons fill shells starting with the lowest energy level before moving outward.
Why Electron Shells Matter
Electron shells determine many important chemical properties of atoms.
The arrangement of electrons influences:
- Chemical bonding
- Reactivity
- Electrical behavior
- Element classification
Atoms are most stable when their outer electron shell is full. This stability explains why certain elements easily form chemical bonds.
The Role of Valence Electrons
The electrons located in the outermost shell of an atom are called valence electrons.
Valence electrons are extremely important because they participate in chemical reactions and bonding between atoms.
Why Valence Electrons Are Important
Valence electrons determine:
- How atoms bond with other atoms
- The types of molecules that form
- Chemical reactivity
- The element’s group in the periodic table
For example:
- Elements with one valence electron tend to lose it easily.
- Elements with seven valence electrons tend to gain one more to complete their outer shell.
This drive toward a full outer shell is known as the octet rule in many atoms.
Electron Shells and the Periodic Table
The structure of electron shells is closely connected to how the periodic table is organized.
Periods and Electron Shells
The rows of the periodic table are called periods, and each period corresponds to the filling of a new electron shell.
Examples:
- Period 1 elements fill the first shell
- Period 2 elements fill the second shell
- Period 3 elements fill the third shell
As atoms move across a period, electrons are added to the same shell.
Groups and Valence Electrons
The columns of the periodic table are called groups.
Elements within the same group often have the same number of valence electrons, which means they share similar chemical properties.
Examples include:
- Alkali metals with one valence electron
- Halogens with seven valence electrons
- Noble gases with full outer shells
Because noble gases have full outer shells, they are generally chemically stable and unreactive.
How Electrons Move Between Shells
Electrons can sometimes move between energy levels.
This occurs when atoms:
- Absorb energy
- Release energy
Absorbing Energy
When an electron absorbs energy, it can jump to a higher electron shell farther from the nucleus.
Releasing Energy
When an electron returns to a lower energy shell, it releases energy in the form of light or radiation.
This behavior explains phenomena such as:
- Atomic emission spectra
- Light produced in neon signs
- Energy transfer in atoms
Subshells and Orbitals
Within each electron shell, electrons are further arranged into smaller regions called subshells and orbitals.
Although shells represent the general energy level, subshells help describe the more detailed arrangement of electrons.
Common subshell types include:
- s subshell
- p subshell
- d subshell
- f subshell
These structures help explain the complex patterns of electron distribution across the periodic table.
Key Takeaways
Electron shells describe how electrons are arranged around the nucleus of an atom.
Important points to remember:
- Electron shells are energy levels surrounding the nucleus
- Each shell holds a maximum number of electrons
- Electrons fill lower shells before higher shells
- Valence electrons in the outer shell control chemical behavior
- Electron shell structure helps explain patterns in the periodic table
Understanding electron shells allows scientists to predict how atoms bond, react, and form the vast variety of substances found in the universe.