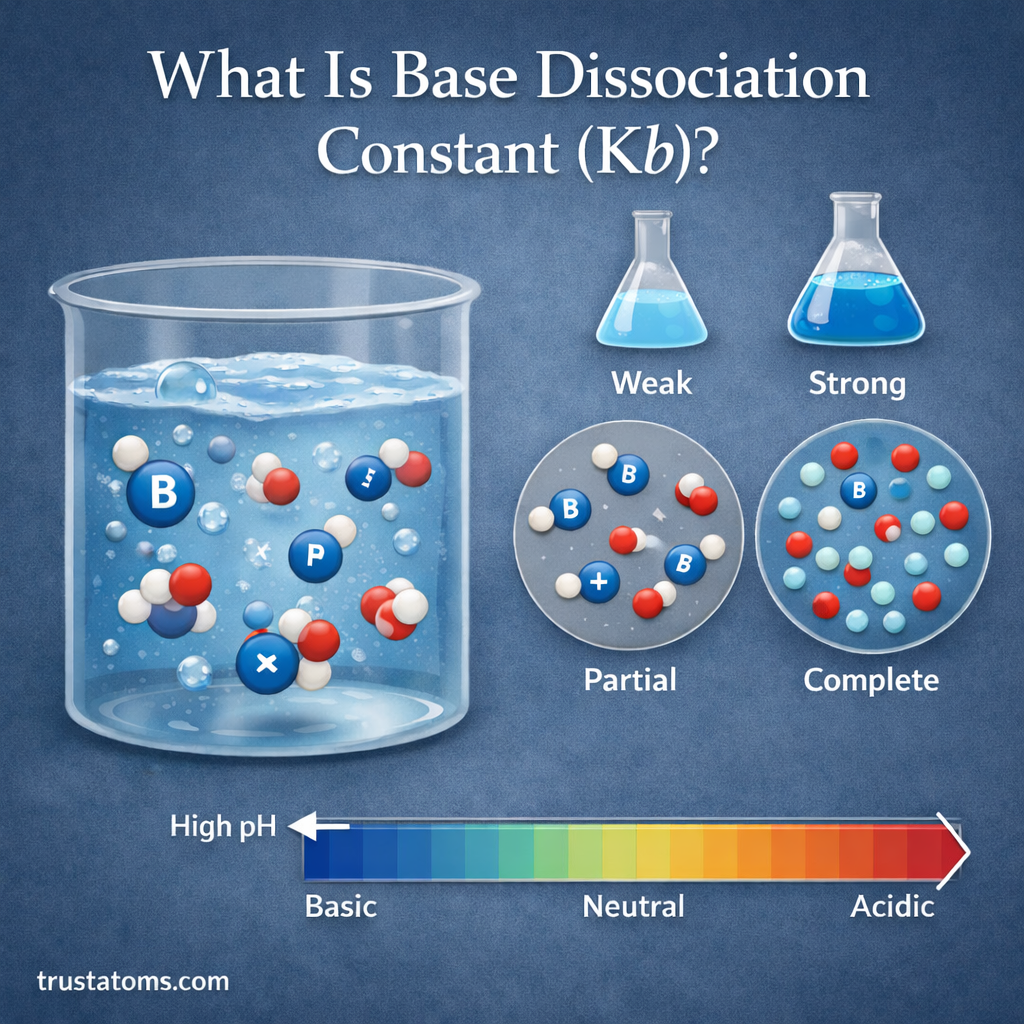
In chemistry, understanding how substances behave in water is essential for studying reactions, biological systems, and industrial processes. One important concept used to measure the strength of a base is the base dissociation constant, commonly written as Kb.
The base dissociation constant helps chemists determine how strongly a base reacts with water to produce hydroxide ions. The larger the Kb value, the stronger the base.
This guide explains what the base dissociation constant is, how it works, how it is calculated, and why it matters in chemistry.
Understanding the Base Dissociation Constant (Kb)
The base dissociation constant (Kb) is a numerical value that measures the strength of a base when it dissolves in water.
When a base dissolves, it reacts with water and produces hydroxide ions (OH⁻). The base dissociation constant describes the equilibrium between the dissolved base and the ions it produces.
In simple terms:
- Large Kb value → strong base
- Small Kb value → weak base
Strong bases dissociate almost completely in water, while weak bases only partially dissociate.
For weak bases, the equilibrium between reactants and products can be expressed using the base dissociation constant.
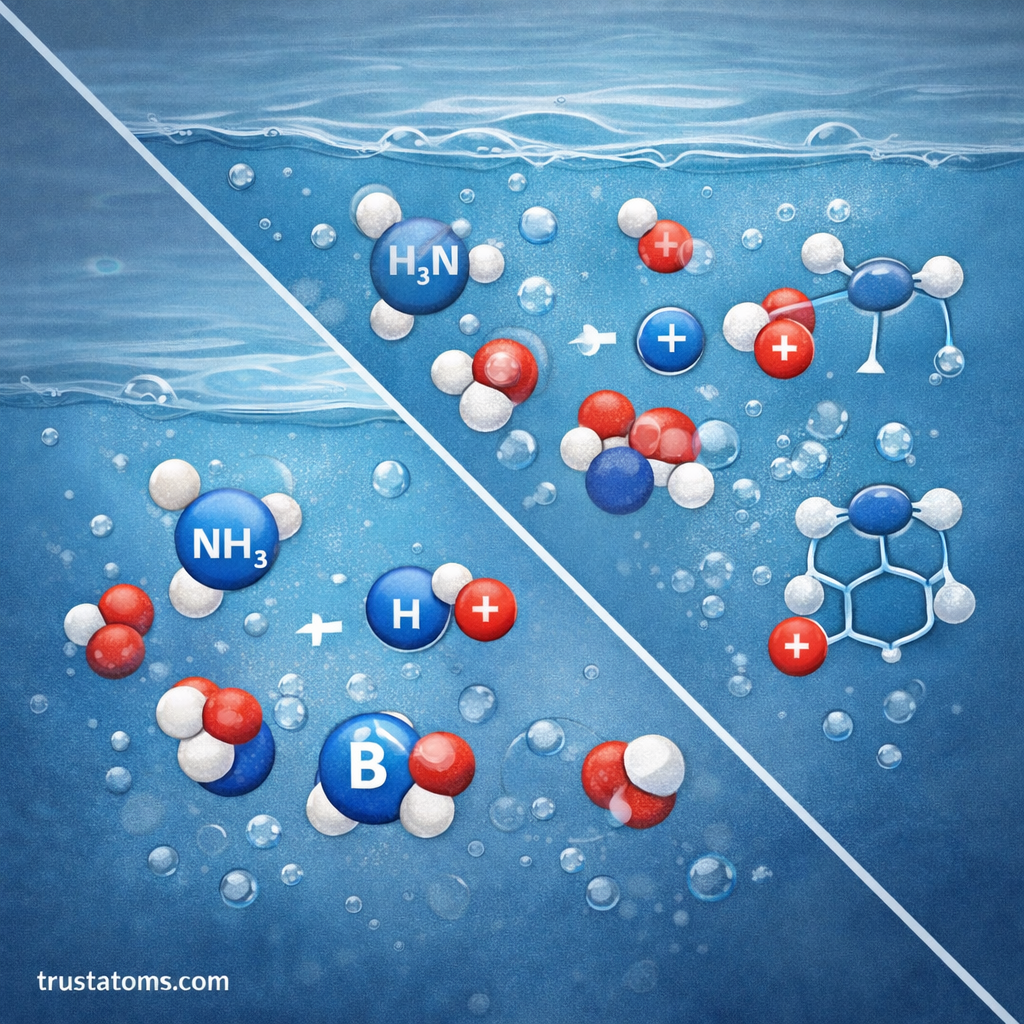
How Bases React in Water
When a base dissolves in water, it typically accepts a proton (H⁺) from a water molecule. This process produces a conjugate acid and hydroxide ions (OH⁻).
For a general base B, the reaction looks like this:
B + H₂O ⇌ BH⁺ + OH⁻
Where:
- B = base
- H₂O = water
- BH⁺ = conjugate acid
- OH⁻ = hydroxide ion
This reaction does not always go completely to the right. Instead, it reaches a dynamic equilibrium, meaning both the forward and reverse reactions occur continuously.
The Kb value quantifies where that equilibrium lies.
The Kb Expression
The base dissociation constant is written using an equilibrium expression derived from the reaction.
For the reaction:
B + H₂O ⇌ BH⁺ + OH⁻
The base dissociation constant is:
Kb = [BH⁺][OH⁻] / [B]
In this expression:
- [BH⁺] = concentration of the conjugate acid
- [OH⁻] = concentration of hydroxide ions
- [B] = concentration of the base
Water is not included in the equation because its concentration remains nearly constant in dilute aqueous solutions.
What the Kb Value Tells Us
The Kb value helps chemists understand how much a base dissociates in water.
Large Kb Value
A large Kb means the base reacts strongly with water.
This results in:
- Higher hydroxide ion concentration
- More complete dissociation
- Greater basicity
Examples include stronger bases such as ammonia derivatives.
Small Kb Value
A small Kb indicates a weak base that only partially reacts with water.
This results in:
- Lower hydroxide ion concentration
- Limited dissociation
- Mild basic properties
Many organic amines and nitrogen-containing compounds fall into this category.
Relationship Between Kb and Base Strength
Base strength directly depends on the Kb value.
The relationship can be summarized as:
| Kb Value | Base Strength |
|---|---|
| Large Kb | Stronger base |
| Small Kb | Weaker base |
However, in practice, chemists often work with weak bases, because strong bases dissociate nearly completely and their equilibrium constants become extremely large.
Relationship Between Kb and pKb
Just like acids have pKa, bases have pKb.
The pKb is simply the logarithmic form of the base dissociation constant.
pKb = −log(Kb)
This relationship helps simplify calculations and comparisons.
Important points:
- Small pKb → strong base
- Large pKb → weak base
Because pKb uses a logarithmic scale, small changes represent large differences in base strength.
Relationship Between Kb and Ka
Every base has a conjugate acid, and their equilibrium constants are related.
The relationship between Kb and Ka is:
Ka × Kb = Kw
Where:
- Ka = acid dissociation constant
- Kb = base dissociation constant
- Kw = ion product of water (1 × 10⁻¹⁴ at 25°C)
This means:
- A strong base has a weak conjugate acid
- A weak base has a stronger conjugate acid
For example:
- Ammonia (NH₃) is a weak base
- Its conjugate acid, ammonium (NH₄⁺), is a weak acid
Understanding this relationship helps chemists analyze acid–base equilibria.
Example: Ammonia as a Weak Base
Ammonia (NH₃) is a common example used to explain Kb.
When ammonia dissolves in water, it reacts as follows:
NH₃ + H₂O ⇌ NH₄⁺ + OH⁻
For ammonia:
Kb ≈ 1.8 × 10⁻⁵
This relatively small Kb value shows that ammonia is a weak base that only partially dissociates in water.
As a result:
- Some ammonia molecules remain unreacted
- A limited number of hydroxide ions are produced
- The solution becomes moderately basic
Why the Base Dissociation Constant Matters
The base dissociation constant is important across many areas of chemistry.
1. Predicting pH of Basic Solutions
Kb helps determine the hydroxide ion concentration, which directly affects the pH of a solution.
Chemists use Kb calculations to estimate how basic a solution will become.
2. Studying Chemical Equilibrium
Kb values help explain how reactions shift between reactants and products.
This is essential for:
- reaction prediction
- solution chemistry
- equilibrium analysis
3. Understanding Biological Systems
Many biological molecules behave as weak bases or acids.
Kb values help explain:
- enzyme function
- cellular buffering systems
- biochemical reactions
4. Industrial and Environmental Chemistry
Base dissociation constants are important in:
- water treatment
- pharmaceutical chemistry
- environmental pH control
- chemical manufacturing
Common Weak Bases and Their Kb Values
Several common substances act as weak bases in water.
Examples include:
| Base | Approximate Kb |
|---|---|
| Ammonia (NH₃) | 1.8 × 10⁻⁵ |
| Methylamine (CH₃NH₂) | 4.4 × 10⁻⁴ |
| Pyridine (C₅H₅N) | 1.7 × 10⁻⁹ |
These values demonstrate how base strength can vary widely depending on molecular structure.
Key Takeaways
The base dissociation constant is a central concept in acid–base chemistry.
Important points to remember:
- Kb measures the strength of a base in water
- Large Kb values indicate stronger bases
- Small Kb values indicate weaker bases
- Kb relates to pKb through a logarithmic scale
- Kb and Ka are connected through the ion product of water
By understanding Kb, chemists can predict solution behavior, analyze chemical equilibria, and better understand the chemistry of both laboratory and biological systems.