Gas exchange in the lungs depends on a fundamental principle of physics: gases move from areas of higher pressure to areas of lower pressure. In the respiratory system, this process is driven by differences in partial pressure, allowing oxygen to enter the blood and carbon dioxide to leave it.
Understanding partial pressure and gas diffusion is essential for explaining how breathing supports life at a cellular level.
What Is Partial Pressure?
Partial pressure refers to the pressure exerted by a single gas within a mixture of gases.
In the air we breathe:
- Oxygen (O₂) makes up about 21%
- Nitrogen (N₂) makes up about 78%
- Carbon dioxide (CO₂) is present in very small amounts
Each gas contributes its own pressure. This individual contribution is called its partial pressure.
Key Idea:
- Higher partial pressure = higher concentration of that gas
- Lower partial pressure = lower concentration
Gases naturally move from areas of higher partial pressure to lower partial pressure.
What Is Gas Diffusion?
Diffusion is the movement of molecules from an area of higher concentration (or pressure) to an area of lower concentration.
In the lungs:
- Oxygen diffuses from the alveoli into the blood
- Carbon dioxide diffuses from the blood into the alveoli
This exchange happens passively—no energy is required.
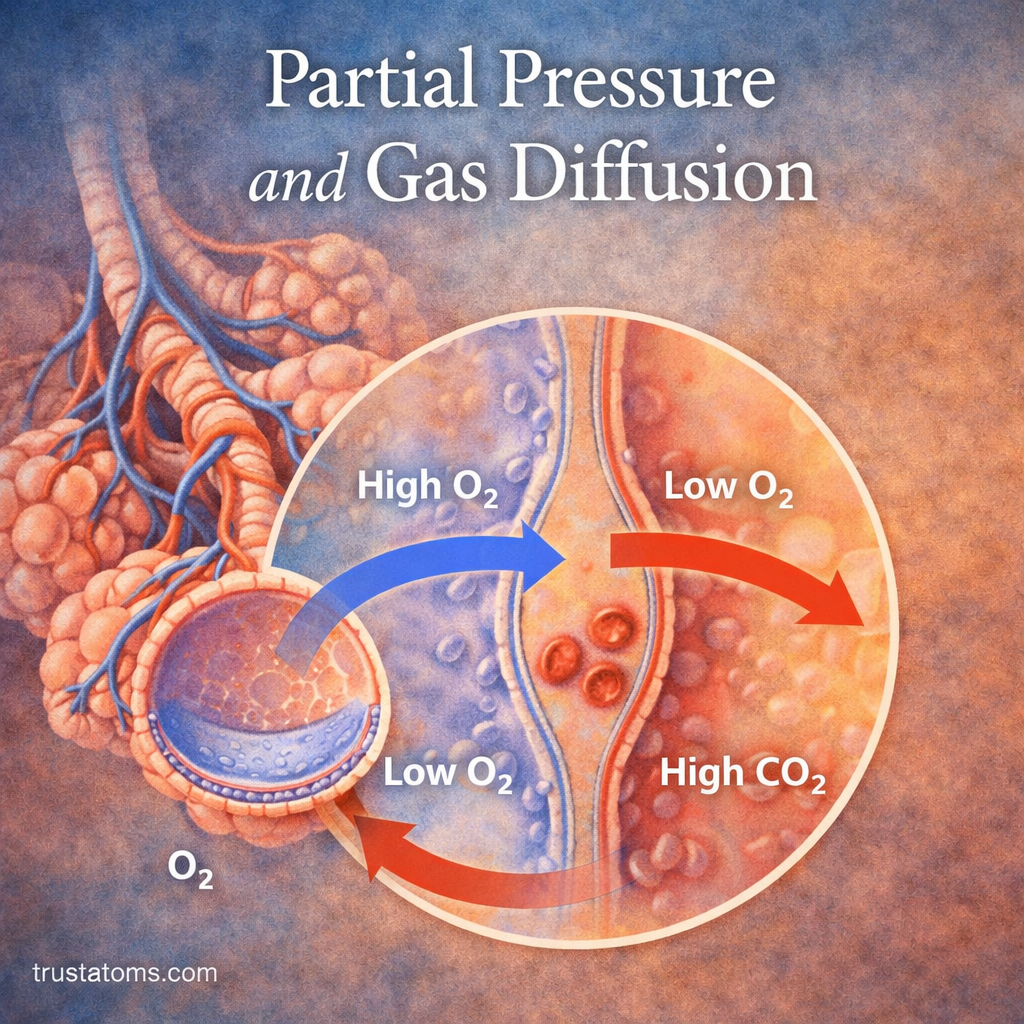
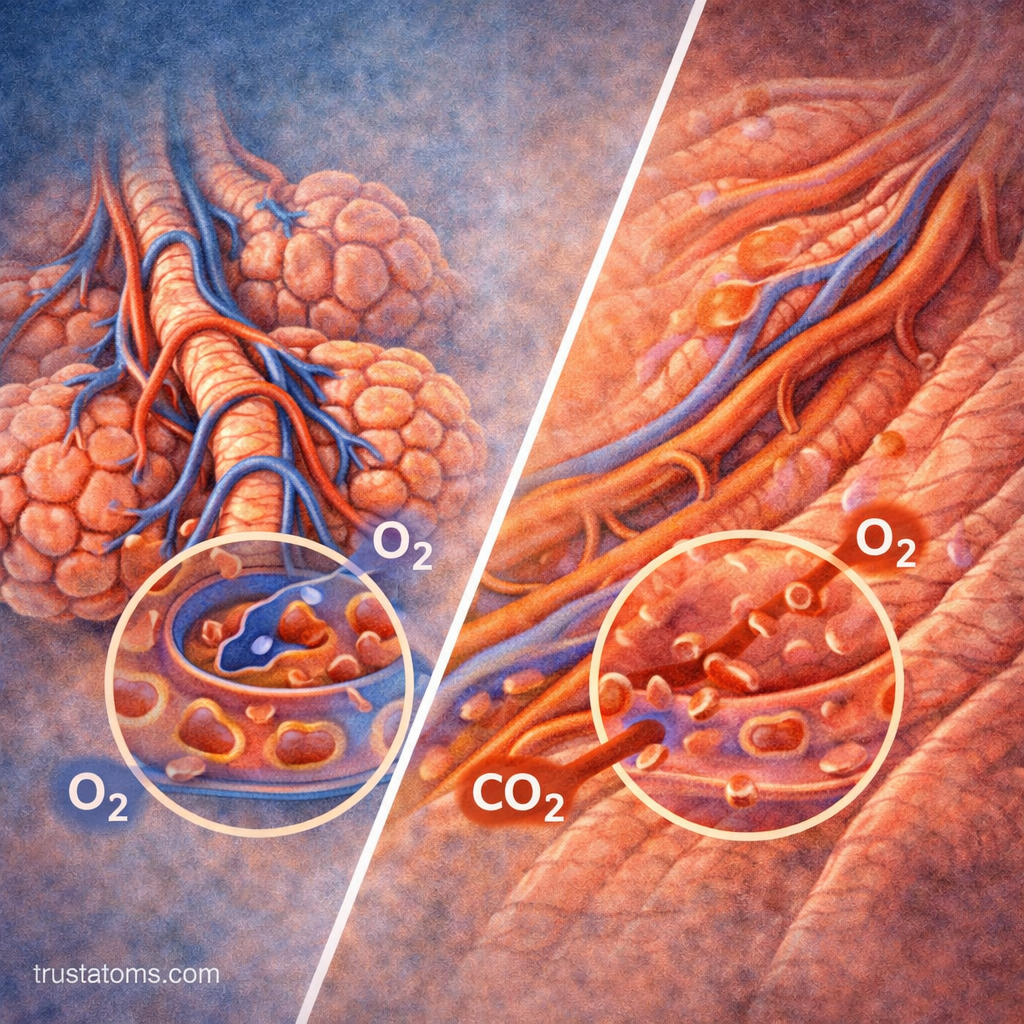
How Partial Pressure Drives Gas Exchange
Gas exchange in the lungs is entirely dependent on partial pressure differences between the alveoli and the blood.
Oxygen Movement
- Oxygen partial pressure is higher in the alveoli
- Oxygen partial pressure is lower in deoxygenated blood
- Oxygen diffuses into the blood
Carbon Dioxide Movement
- Carbon dioxide partial pressure is higher in the blood
- Carbon dioxide partial pressure is lower in the alveoli
- Carbon dioxide diffuses into the alveoli
This continuous gradient ensures efficient gas exchange with every breath.
The Alveolar-Capillary Membrane
Gas diffusion occurs across a very thin barrier called the alveolar-capillary membrane.
This membrane includes:
- Alveolar wall (epithelium)
- Capillary wall (endothelium)
- A thin shared basement membrane
Because it is extremely thin, gases can diffuse quickly and efficiently.
Factors That Affect Gas Diffusion
Several factors influence how easily gases move across the alveoli.
1. Surface Area
- Larger surface area increases diffusion
- Reduced surface area slows gas exchange
2. Membrane Thickness
- Thinner membranes allow faster diffusion
- Thickened membranes slow the process
3. Partial Pressure Gradient
- Greater difference = faster diffusion
- Smaller difference = slower diffusion
4. Gas Properties
- Oxygen and carbon dioxide diffuse at different rates
- Carbon dioxide diffuses more easily than oxygen
Partial Pressure in the Body
Partial pressure differences are not limited to the lungs—they also occur in body tissues.
In the Tissues:
- Oxygen moves from blood (higher pressure) → cells (lower pressure)
- Carbon dioxide moves from cells (higher pressure) → blood (lower pressure)
This ensures that:
- Cells receive oxygen for energy production
- Carbon dioxide waste is removed
Ventilation and Perfusion Balance
Efficient gas exchange depends on matching:
- Ventilation (air reaching alveoli)
- Perfusion (blood reaching alveoli)
If this balance is disrupted:
- Partial pressure gradients weaken
- Gas exchange becomes less efficient
This is known as ventilation-perfusion (V/Q) mismatch.
Conditions That Affect Gas Diffusion
Certain conditions can interfere with partial pressure gradients and diffusion.
1. Pulmonary Edema
- Fluid in alveoli increases diffusion distance
- Slows oxygen movement
2. Emphysema
- Reduces alveolar surface area
- Decreases gas exchange efficiency
3. Fibrosis
- Thickens the alveolar membrane
- Impairs diffusion
4. High Altitude
- Lower oxygen partial pressure in air
- Reduces oxygen diffusion into blood
Why Partial Pressure Is Essential for Life
Partial pressure gradients are the driving force behind:
- Oxygen delivery to tissues
- Carbon dioxide removal from the body
Without these gradients:
- Cells would not receive enough oxygen
- Waste gases would accumulate
- Vital processes would fail
Final Thoughts
Partial pressure and gas diffusion are at the core of how the respiratory system functions. By creating pressure differences between the lungs, blood, and tissues, the body ensures a constant flow of oxygen in and carbon dioxide out.
This simple yet powerful mechanism supports every cell in the body, making it one of the most essential processes for life.