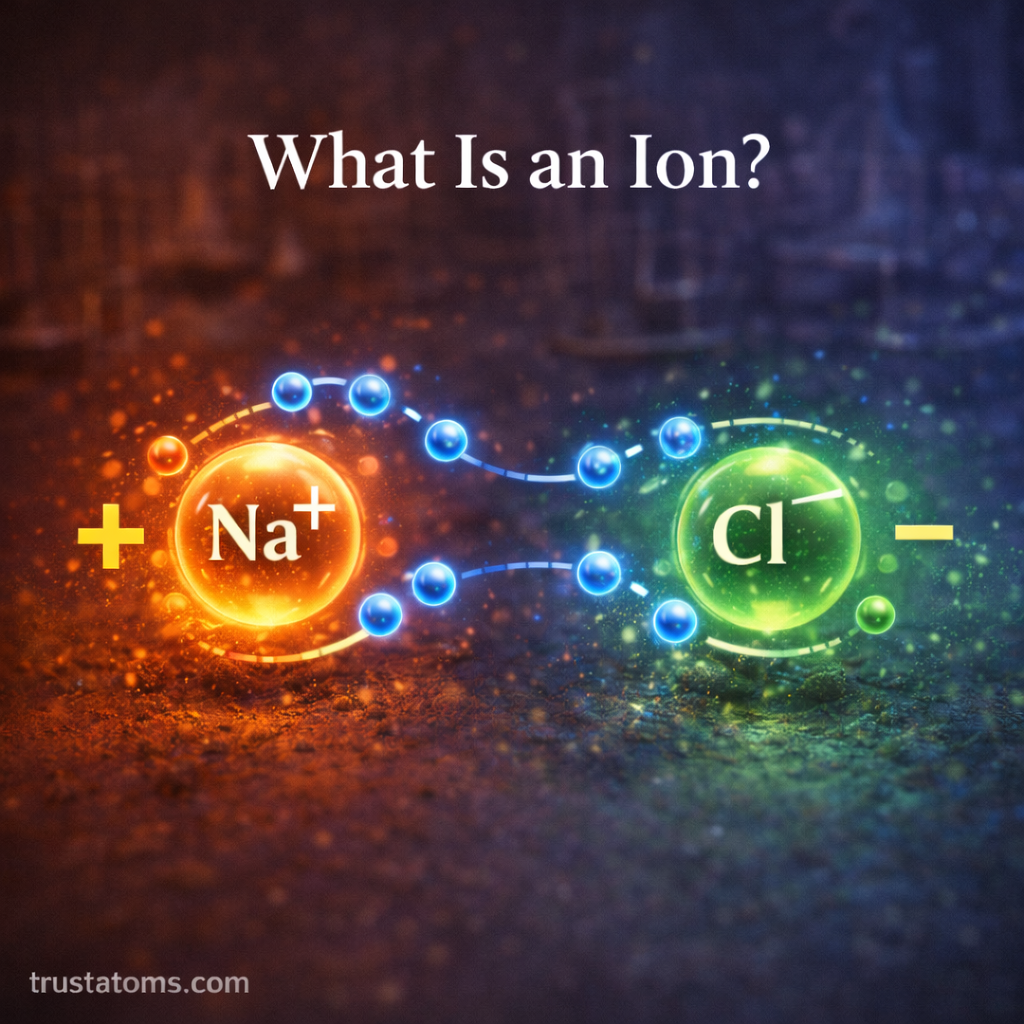
An ion is an atom or molecule that carries an electric charge because it has gained or lost one or more electrons. In normal conditions, atoms are electrically neutral, meaning they have equal numbers of protons and electrons. When that balance changes, the atom becomes an ion.
Ions play a critical role in chemistry, biology, and physics. They are involved in chemical reactions, electricity in batteries, nerve signals in the body, and the formation of many compounds found in nature.
Ion Definition
An ion is a particle formed when an atom or molecule gains or loses electrons, giving it a positive or negative electrical charge.
This change happens because electrons are relatively easy for atoms to lose or gain during chemical interactions.
There are two main types of ions:
- Cations (positively charged ions)
- Anions (negatively charged ions)
The type of ion depends on whether electrons are lost or gained.
How Ions Form
Ions form when atoms transfer electrons during chemical reactions.
Atoms tend to seek stability by achieving a full outer electron shell. To reach that stable state, atoms may:
- Lose electrons
- Gain electrons
- Share electrons with other atoms
When electrons are transferred rather than shared, ions are created.
Example
A sodium atom can lose one electron. When this happens:
- It now has more protons than electrons
- It becomes positively charged
- It forms a sodium ion
Similarly, a chlorine atom may gain an electron, becoming negatively charged and forming a chloride ion.
Types of Ions
Cations
Cations are ions with a positive charge.
They form when an atom loses electrons. Because electrons carry negative charge, losing them leaves the atom with a net positive charge.
Common examples include:
- Sodium ion
- Calcium ion
- Potassium ion
- Hydrogen ion
Many metals commonly form cations.
Anions
Anions are ions with a negative charge.
They form when an atom gains electrons. The extra electrons increase the atom’s negative charge.
Common examples include:
- Chloride ion
- Oxide ion
- Nitrate ion
- Sulfate ion
Nonmetals frequently form anions.
Monatomic vs Polyatomic Ions
Ions can consist of either a single atom or a group of atoms.
Monatomic Ions
Monatomic ions are formed from a single atom that has gained or lost electrons.
Examples include:
- Sodium ion
- Magnesium ion
- Chloride ion
- Oxide ion
These ions come directly from individual elements.
Polyatomic Ions
Polyatomic ions are made of two or more atoms bonded together that collectively carry a charge.
Examples include:
- Nitrate ion
- Carbonate ion
- Ammonium ion
- Phosphate ion
Even though they contain multiple atoms, they behave as a single charged unit during chemical reactions.
Why Ions Are Important
Ions are essential for many chemical and biological processes.
They play key roles in:
- Formation of ionic compounds
- Electrical conductivity in solutions
- Battery and electrochemical reactions
- Biological nerve signals
- Muscle contractions
- Cellular communication
Without ions, many life-sustaining processes would not function.
Ions in Ionic Bonds
Ions are responsible for ionic bonding, a type of chemical bond formed by electrostatic attraction between oppositely charged ions.
In ionic bonding:
- One atom loses electrons and becomes a cation
- Another atom gains electrons and becomes an anion
- Opposite charges attract, forming a stable compound
A common example is table salt.
Sodium loses an electron to become a sodium ion, while chlorine gains that electron to become a chloride ion. The two ions attract each other and form sodium chloride.
Ions in Everyday Life

Ions are everywhere in daily life, often in ways people don’t realize.
Examples include:
Electrolytes in the Body
Electrolytes such as sodium, potassium, calcium, and chloride ions help regulate:
- Hydration
- Nerve signals
- Muscle movement
- Blood pressure
Sports drinks often contain these ions to replenish lost electrolytes.
Batteries
Batteries work by moving ions through a chemical medium while electrons flow through an external circuit.
This movement of charged particles generates electrical energy.
Water Treatment
Ions are involved in processes such as water softening and purification, where unwanted ions are removed or replaced.
How to Identify an Ion
You can often recognize ions by their electrical charge notation.
Chemists write ions using a superscript indicating the charge.
Examples include:
- Na⁺ (sodium ion)
- Ca²⁺ (calcium ion)
- Cl⁻ (chloride ion)
- SO₄²⁻ (sulfate ion)
The superscript shows how many electrons were gained or lost.
Ions vs Neutral Atoms
The difference between ions and neutral atoms comes down to electrical charge.
Neutral atom:
- Equal number of protons and electrons
- No overall charge
Ion:
- Unequal number of protons and electrons
- Carries a positive or negative charge
This change in charge significantly affects how atoms interact in chemical reactions.
Final Thoughts
An ion is an atom or molecule that carries an electric charge because it has gained or lost electrons. These charged particles are fundamental to chemistry, helping form compounds, conduct electricity, and drive essential biological processes.
From table salt to nerve impulses and batteries, ions play a vital role in the chemical systems that shape both everyday life and advanced technology.