An isotope is a form of an element that has the same number of protons but a different number of neutrons in its atomic nucleus. Because isotopes belong to the same element, they share the same atomic number and similar chemical behavior, but they can differ in mass and physical properties.
Isotopes are important in many areas of science, including chemistry, physics, medicine, archaeology, and environmental research.
Isotope Definition
An isotope is an atom of the same element that contains:
- The same number of protons
- A different number of neutrons
Because protons determine the element, changing the number of neutrons does not create a new element. Instead, it creates a different version of that element.
For example, carbon atoms always contain 6 protons, but they can have different numbers of neutrons. These variations are carbon isotopes.
Why Isotopes Exist
Atoms contain three main subatomic particles:
- Protons (positive charge)
- Neutrons (no charge)
- Electrons (negative charge)
The number of protons defines the element. However, the number of neutrons can vary.
When atoms of the same element contain different numbers of neutrons, they become isotopes.
These changes affect the mass of the atom, which is why isotopes have different atomic masses.
Common Examples of Isotopes
Many elements have multiple isotopes. Some are stable, while others are radioactive.
Carbon Isotopes
Carbon has three well-known isotopes:
- Carbon-12 (6 protons, 6 neutrons)
- Carbon-13 (6 protons, 7 neutrons)
- Carbon-14 (6 protons, 8 neutrons)
Carbon-12 and carbon-13 are stable, while carbon-14 is radioactive and used in radiocarbon dating.
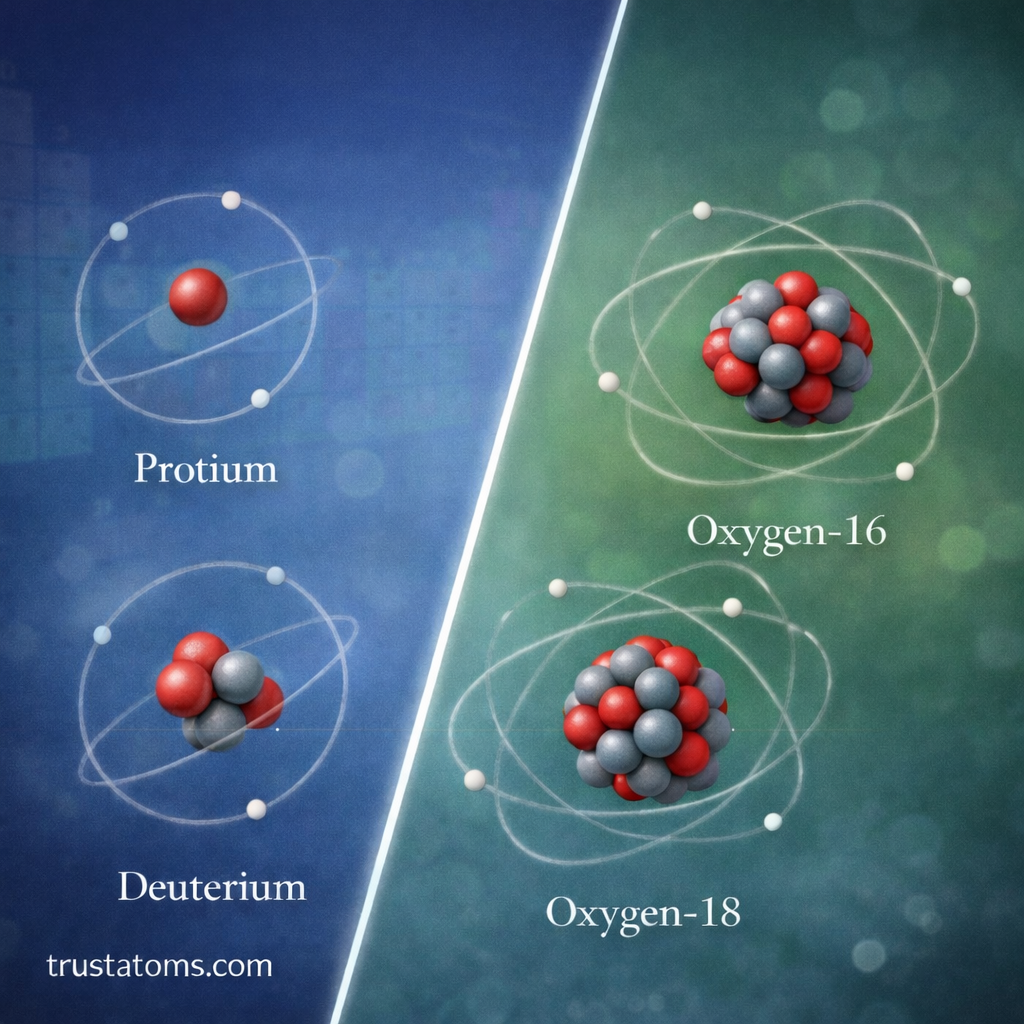
Hydrogen Isotopes
Hydrogen has three important isotopes:
- Protium (1 proton, 0 neutrons)
- Deuterium (1 proton, 1 neutron)
- Tritium (1 proton, 2 neutrons)
These isotopes are used in scientific research, nuclear studies, and energy research.
Stable vs Radioactive Isotopes
Isotopes can be categorized based on their stability.
Stable Isotopes
Stable isotopes do not break down over time. Their nuclei remain unchanged.
Examples include:
- Carbon-12
- Oxygen-16
- Nitrogen-14
Stable isotopes are commonly used in scientific research and environmental studies.
Radioactive Isotopes
Radioactive isotopes are unstable and gradually decay, releasing radiation.
This process is called radioactive decay.
Examples include:
- Carbon-14
- Uranium-235
- Iodine-131
Radioactive isotopes are used in medicine, nuclear power, and scientific dating techniques.
How Isotopes Are Written
Chemists use a standard notation to describe isotopes.
The isotope name usually includes the element name followed by its mass number.
For example:
- Carbon-14
- Oxygen-18
- Uranium-235
The mass number equals:
Protons + Neutrons
This number helps distinguish between different isotopes of the same element.
Why Isotopes Matter in Science
Isotopes are incredibly useful tools for scientists. They help researchers study processes that would otherwise be difficult to observe.
Important applications include:
- Medical imaging and cancer treatment
- Archaeological dating techniques
- Environmental research
- Nuclear energy production
- Chemical and biological tracing
Isotopes allow scientists to track substances and understand how matter changes over time.
Isotopes in Medicine
Radioactive isotopes are widely used in modern medicine.
Doctors use isotopes for:
- Diagnostic imaging (PET scans and medical tracers)
- Cancer radiation therapy
- Studying organ function
For example, iodine-131 is often used to treat thyroid disorders.
These medical uses rely on the controlled radiation emitted during radioactive decay.
Isotopes in Archaeology and Earth Science
One of the most famous uses of isotopes is radiocarbon dating.
Scientists measure the remaining carbon-14 in organic materials to estimate how long ago an organism died.
This technique has helped determine the age of:
- Ancient fossils
- Archaeological artifacts
- Historical remains
Other isotopes are used to study climate history, ocean circulation, and geological processes.
Isotopes vs Elements
It is important to distinguish between isotopes and elements.
Element:
- Defined by the number of protons
- All atoms of the element share this number
Isotope:
- Same number of protons
- Different number of neutrons
- Different atomic mass
This means all isotopes of an element behave chemically in similar ways but may vary in stability and mass.
Final Thoughts
An isotope is a version of an element that contains the same number of protons but a different number of neutrons. These variations change the mass of the atom while keeping its chemical identity the same.
Isotopes play a crucial role in many scientific fields, from medical diagnostics to archaeological dating and nuclear energy. By studying isotopes, scientists gain valuable insight into the structure of matter and the processes shaping our world.