A proton is one of the fundamental particles that make up atoms. Along with neutrons and electrons, protons are essential components of all matter in the universe. These tiny particles play a critical role in determining the identity of every chemical element.
Understanding protons helps explain atomic structure, chemical reactions, and why different elements behave the way they do.
The Basic Definition of a Proton
A proton is a positively charged subatomic particle located in the nucleus of an atom.
The nucleus sits at the center of an atom and contains both protons and neutrons. Electrons move around the nucleus in regions called electron clouds or orbitals.
Key characteristics of protons include:
- Positive electrical charge
- Located in the atomic nucleus
- Much heavier than electrons
- Essential for determining the element type
Because protons carry a positive charge, they attract negatively charged electrons and help keep atoms stable.
Where Protons Are Located in an Atom
Atoms consist of three primary subatomic particles:
- Protons
- Neutrons
- Electrons
Atomic Structure Overview
- Protons are found in the nucleus and carry a positive charge.
- Neutrons are also located in the nucleus and have no electrical charge.
- Electrons move around the nucleus and carry a negative charge.
The nucleus is extremely small compared to the overall size of the atom, yet it contains nearly all of the atom’s mass.
Why Protons Determine the Element
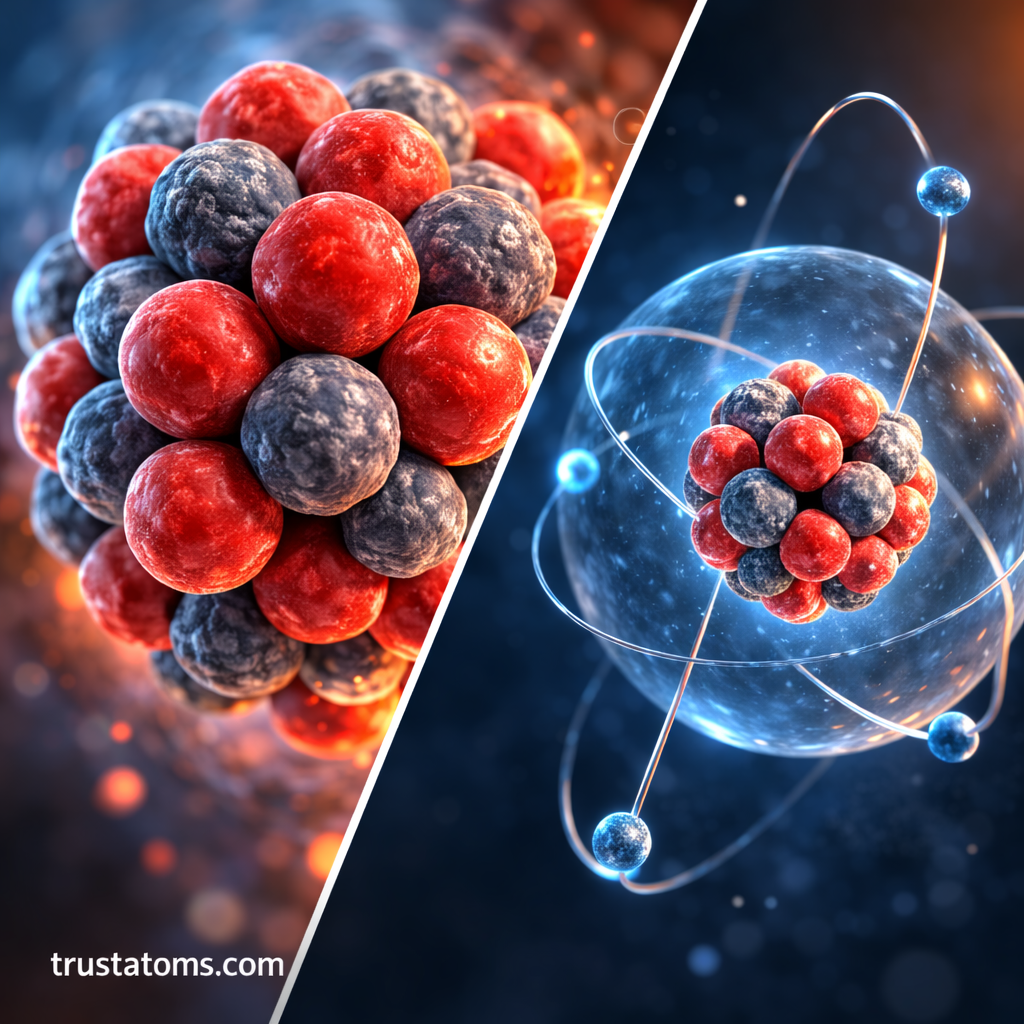
One of the most important roles of protons is determining the atomic number of an element.
The atomic number is simply the number of protons in an atom’s nucleus.
Examples of Atomic Numbers
- Hydrogen → 1 proton
- Carbon → 6 protons
- Oxygen → 8 protons
- Gold → 79 protons
If the number of protons changes, the atom becomes a completely different element.
For example:
- An atom with 6 protons is carbon
- An atom with 7 protons is nitrogen
This is why protons define the identity of each chemical element.
Properties of Protons
Protons have several important physical and electrical properties.
1. Positive Electric Charge
A proton carries a +1 electric charge. This positive charge balances the negative charge of electrons in neutral atoms.
When the number of protons equals the number of electrons, the atom has no overall electrical charge.
2. Significant Mass
Protons are much heavier than electrons.
In fact, a proton’s mass is about 1,836 times greater than the mass of an electron. Because of this, most of an atom’s mass comes from protons and neutrons inside the nucleus.
3. Stability Within the Nucleus
Protons remain tightly bound inside the atomic nucleus by powerful nuclear forces. These forces help hold the nucleus together despite the repulsion between positively charged protons.
Protons and Atomic Stability
Atoms remain stable because of the balance between protons, neutrons, and electrons.
Forces Inside the Atom
Several forces influence atomic stability:
- Electromagnetic attraction
- Opposite charges attract
- Positive protons attract negative electrons
- Electromagnetic repulsion
- Protons repel other protons because they have the same charge
- Strong nuclear force
- A powerful force that holds protons and neutrons together inside the nucleus
Without the strong nuclear force, the positive charges of protons would push the nucleus apart.
The Discovery of the Proton
The proton was discovered in the early 20th century through studies of atomic structure.
Ernest Rutherford’s Contribution
Physicist Ernest Rutherford identified the proton in 1917 during experiments that involved bombarding nitrogen gas with alpha particles.
These experiments revealed that hydrogen nuclei were being produced, leading Rutherford to conclude that hydrogen nuclei were fundamental components of atomic nuclei. These particles were later named protons.
Rutherford’s work helped establish the modern understanding of atomic structure.
The Role of Protons in Chemistry
Protons influence many chemical properties and behaviors of elements.
Chemists rely on the number of protons in an atom to determine:
- The element’s identity
- Its position on the periodic table
- Its chemical behavior
- Its bonding tendencies
Because protons determine the atomic number, they also determine how electrons are arranged around the nucleus.
This electron arrangement influences how atoms interact with one another during chemical reactions.
Protons and the Periodic Table
The periodic table is organized by increasing atomic number, which corresponds directly to the number of protons in each element.
This organization allows scientists to predict properties such as:
- Chemical reactivity
- Atomic size
- Bonding behavior
- Ion formation
Elements with similar electron structures often appear in the same groups, which explains why they have similar chemical properties.
Key Takeaways
Protons are fundamental particles that define the identity and structure of atoms.
Important facts about protons include:
- Protons are positively charged subatomic particles
- They are located in the nucleus of an atom
- The number of protons determines the atomic number
- Changing the number of protons changes the element itself
- Protons contribute significantly to an atom’s mass
Understanding protons is essential for studying chemistry, physics, and the structure of matter.