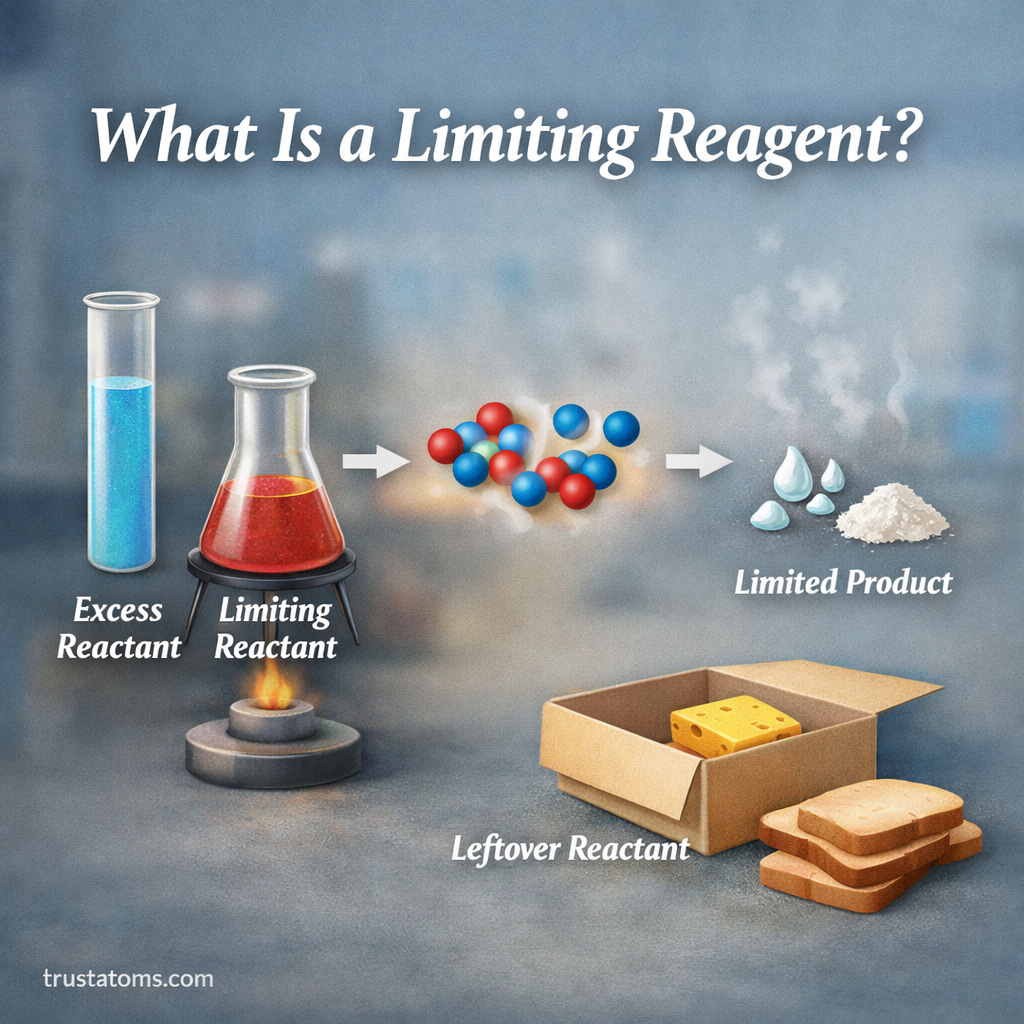
In many chemical reactions, substances combine in specific ratios based on a balanced chemical equation. However, the reactants available in the laboratory or in nature are not always present in exactly those ratios.
The limiting reagent (also called the limiting reactant) is the substance that runs out first during a chemical reaction, preventing more product from forming. Because it is consumed completely, it determines the maximum amount of product the reaction can produce.
Understanding limiting reagents is essential in chemistry because it helps scientists predict reaction yields, reduce waste, and optimize industrial chemical processes.
The Basic Idea of a Limiting Reagent

A limiting reagent is similar to a limiting ingredient in cooking.
Imagine making sandwiches:
- Each sandwich requires 2 slices of bread and 1 slice of cheese
- If you have 10 slices of bread but only 3 slices of cheese, you can only make 3 sandwiches
Even though you have extra bread, the cheese limits how many sandwiches you can make.
Chemical reactions work the same way. The reactant that runs out first limits how much product can form.
Why Limiting Reagents Matter in Chemistry
Identifying the limiting reagent allows chemists to:
- Predict how much product will form
- Determine how much excess reactant remains
- Improve efficiency in industrial reactions
- Reduce waste in chemical manufacturing
- Calculate percent yield in experiments
Without determining the limiting reagent, it would be impossible to accurately calculate reaction outcomes.
Limiting Reagent in a Chemical Reaction
Chemical reactions follow stoichiometry, meaning reactants combine in fixed mole ratios defined by the balanced equation.
Consider this reaction:
2H₂ + O₂ → 2H₂O
This equation tells us:
- 2 molecules of hydrogen react with 1 molecule of oxygen
- The reaction produces 2 molecules of water
If we mix:
- 4 molecules of hydrogen
- 1 molecule of oxygen
Only 2 molecules of hydrogen can react, because the equation requires a 2:1 ratio.
Oxygen runs out first, so oxygen is the limiting reagent, and some hydrogen remains unreacted.
Steps to Identify the Limiting Reagent
Chemists use a systematic method to determine which reactant limits a reaction.
1. Write and Balance the Chemical Equation
A balanced equation shows the correct ratio of reactants.
Example:
N₂ + 3H₂ → 2NH₃
2. Convert Quantities to Moles
Stoichiometry works in moles, so masses must be converted using molar mass.
Example:
Mass ÷ Molar Mass = Moles
3. Use Mole Ratios from the Equation
Determine how much product each reactant could produce.
4. Identify the Smaller Product Amount
The reactant that produces less product is the limiting reagent.
This reactant will be used up first, stopping the reaction.
Example Calculation
Suppose we react:
- 10 moles of hydrogen (H₂)
- 5 moles of oxygen (O₂)
Balanced equation:
2H₂ + O₂ → 2H₂O
Step 1: Compare mole ratios.
According to the equation:
- 2 moles H₂ react with 1 mole O₂
Step 2: Check oxygen requirement.
If we have 10 moles of H₂, we need:
10 ÷ 2 = 5 moles of O₂
Since we have exactly 5 moles of O₂, neither reactant is limiting. They are present in the exact ratio required.
If we had only 3 moles of O₂, then oxygen would be the limiting reagent.
What Is an Excess Reagent?
The excess reagent is the reactant that remains after the limiting reagent is consumed.
Characteristics of an excess reagent:
- Not completely used up
- Remains after the reaction finishes
- Often intentionally added in excess to ensure the limiting reagent reacts completely
For example, in many industrial reactions, companies deliberately add one reactant in excess to maximize product formation.
Limiting Reagents in Real-World Applications
Limiting reagents play a critical role in many scientific and industrial processes.
Industrial Chemistry
Chemical manufacturing relies on limiting reagent calculations to:
- Control production costs
- Reduce leftover chemicals
- Maximize product yield
Pharmaceuticals
Drug manufacturing requires extremely precise chemical reactions. Identifying the limiting reagent ensures:
- Consistent drug production
- Accurate dosing
- Reduced contamination risks
Environmental Chemistry
Limiting reagent analysis helps scientists understand:
- Pollution reactions in the atmosphere
- Nutrient cycles in ecosystems
- Chemical breakdown of pollutants
Laboratory Experiments
Students and researchers routinely calculate limiting reagents to determine:
- Theoretical yield
- Percent yield
- Reaction efficiency
Common Mistakes When Identifying a Limiting Reagent
Students often make a few common errors when solving limiting reagent problems.
Not Balancing the Equation First
Stoichiometric ratios come from the balanced equation, so an unbalanced equation leads to incorrect answers.
Comparing Mass Instead of Moles
Chemical reactions occur between moles of particles, not grams.
Always convert to moles first.
Ignoring Mole Ratios
Even if one reactant has a smaller mass, it might not be limiting if the reaction ratio requires less of it.
Limiting Reagent vs Theoretical Yield
The limiting reagent directly determines the theoretical yield of a reaction.
Theoretical yield is:
The maximum amount of product that can be formed based on the limiting reagent.
Actual reactions rarely reach 100% efficiency, which is why chemists calculate percent yield by comparing theoretical and actual results.
Final Thoughts
The limiting reagent is a central concept in chemistry because it determines how far a reaction can proceed. By identifying which reactant runs out first, chemists can predict product formation, control industrial reactions, and improve experimental accuracy.
From laboratory experiments to large-scale chemical manufacturing, understanding limiting reagents helps scientists work more efficiently and minimize wasted materials.
Mastering this concept also builds a strong foundation for other chemistry topics such as stoichiometry, reaction yields, and chemical equilibrium.