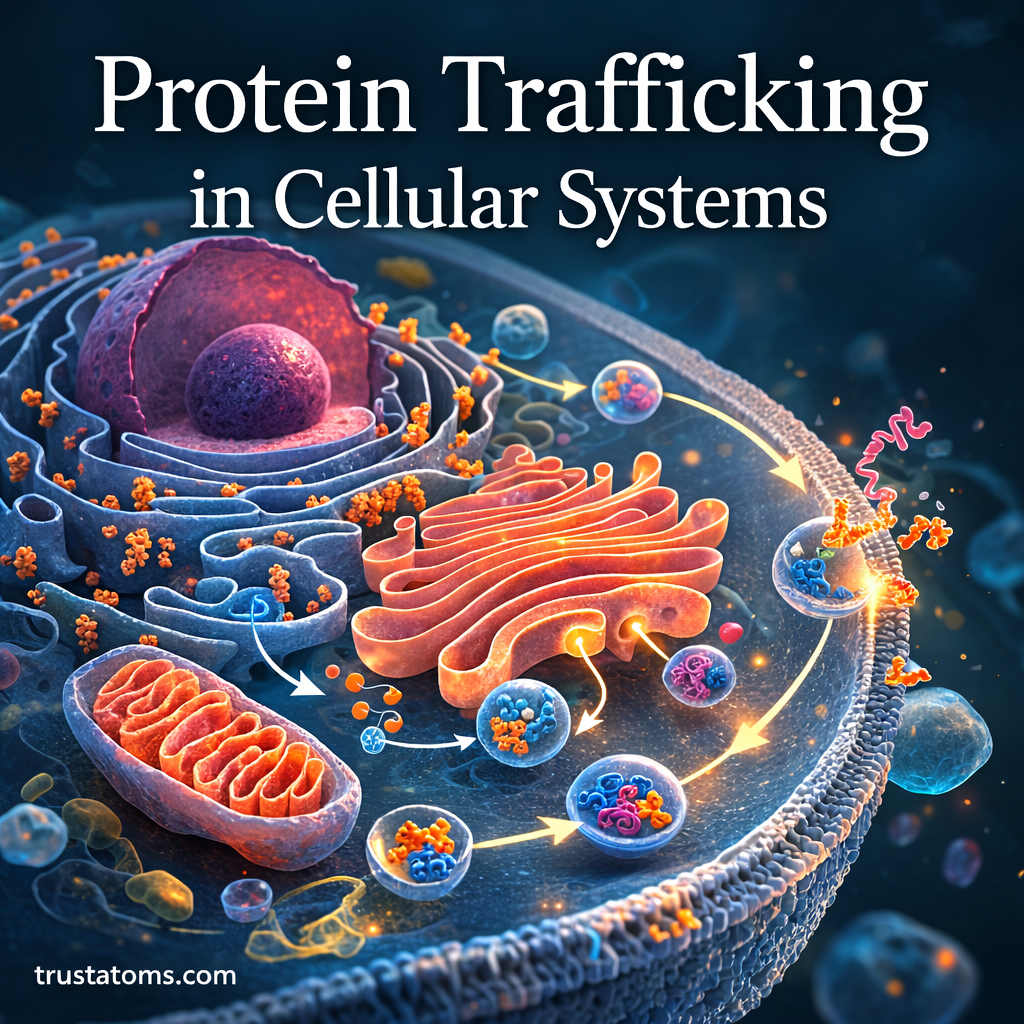
Proteins are essential molecules that perform countless functions inside cells, including catalyzing chemical reactions, transporting substances, regulating genes, and maintaining structural integrity. However, proteins must first reach the correct cellular location in order to function properly. This process is known as protein trafficking.
Protein trafficking refers to the highly organized system that directs newly synthesized proteins to specific destinations within or outside the cell. Cells rely on precise targeting mechanisms, transport pathways, and signaling sequences to ensure proteins reach the correct organelles or cellular compartments.
Understanding protein trafficking is essential for studying cell biology, disease mechanisms, and biotechnology applications because errors in protein transport can lead to severe cellular dysfunction.
What Is Protein Trafficking?
Protein trafficking is the process by which proteins are transported from the site of their synthesis to their final functional location within a cell.
Proteins may be directed to several destinations, including:
- The cell membrane
- Organelles such as mitochondria or lysosomes
- The cytoplasm
- The nucleus
- Outside the cell through secretion
Cells use a complex system of molecular signals and transport pathways to guide proteins through the correct route.
This coordinated movement ensures that proteins arrive where they are needed for proper cellular function.
Where Protein Synthesis Begins
Protein trafficking begins immediately after proteins are synthesized.
Ribosomes and Protein Production
Proteins are produced by ribosomes through a process known as translation, where genetic information encoded in messenger RNA (mRNA) is converted into amino acid chains.
Ribosomes may be located in two places:
- Free in the cytoplasm
- Attached to the rough endoplasmic reticulum (ER)
The location of protein synthesis often determines the protein’s eventual destination.
Free Ribosomes
Proteins synthesized by free ribosomes usually remain within the cytoplasm or are transported to internal organelles.
These proteins commonly function in:
- Metabolic pathways
- Cellular structure
- Nuclear processes
- Mitochondrial activity
Free ribosome proteins typically contain targeting signals that direct them to the correct organelle.
Rough Endoplasmic Reticulum
Proteins synthesized on ribosomes attached to the rough ER are typically destined for:
- Secretion outside the cell
- The cell membrane
- The endomembrane system
During synthesis, these proteins are inserted directly into the ER lumen or membrane.
Signal Sequences and Protein Targeting
Cells use signal sequences, short stretches of amino acids, to guide proteins to their proper destinations.
These sequences act as molecular addresses that determine where proteins should go.
Examples of targeting signals include:
- ER signal peptides
- Nuclear localization signals
- Mitochondrial targeting sequences
- Peroxisomal targeting signals
Specialized cellular machinery recognizes these signals and directs the protein into the appropriate trafficking pathway.
The Endomembrane System and Protein Transport
Many proteins travel through the endomembrane system, a network of interconnected organelles involved in protein processing and transport.
Key components include:
- Endoplasmic reticulum
- Golgi apparatus
- Transport vesicles
- Lysosomes
- Plasma membrane
This system functions as the cell’s internal shipping network.
Protein Processing in the Endoplasmic Reticulum
Once proteins enter the rough ER, they undergo several important modifications.
Protein Folding
Newly synthesized proteins must fold into precise three-dimensional shapes to function correctly.
Special proteins called chaperones assist with this folding process.
Incorrectly folded proteins are usually targeted for degradation to prevent cellular damage.
Chemical Modifications
Proteins may also undergo modifications such as:
- Glycosylation (addition of sugar molecules)
- Formation of disulfide bonds
- Structural adjustments
These modifications often affect protein stability, activity, and transport.
The Role of the Golgi Apparatus
After leaving the ER, proteins are transported to the Golgi apparatus.
The Golgi functions as a sorting and processing center for proteins.
Within the Golgi, proteins may undergo additional modifications, including:
- Further glycosylation
- Structural processing
- Tagging for specific destinations
The Golgi also determines where proteins will be delivered next.
Vesicle-Mediated Transport
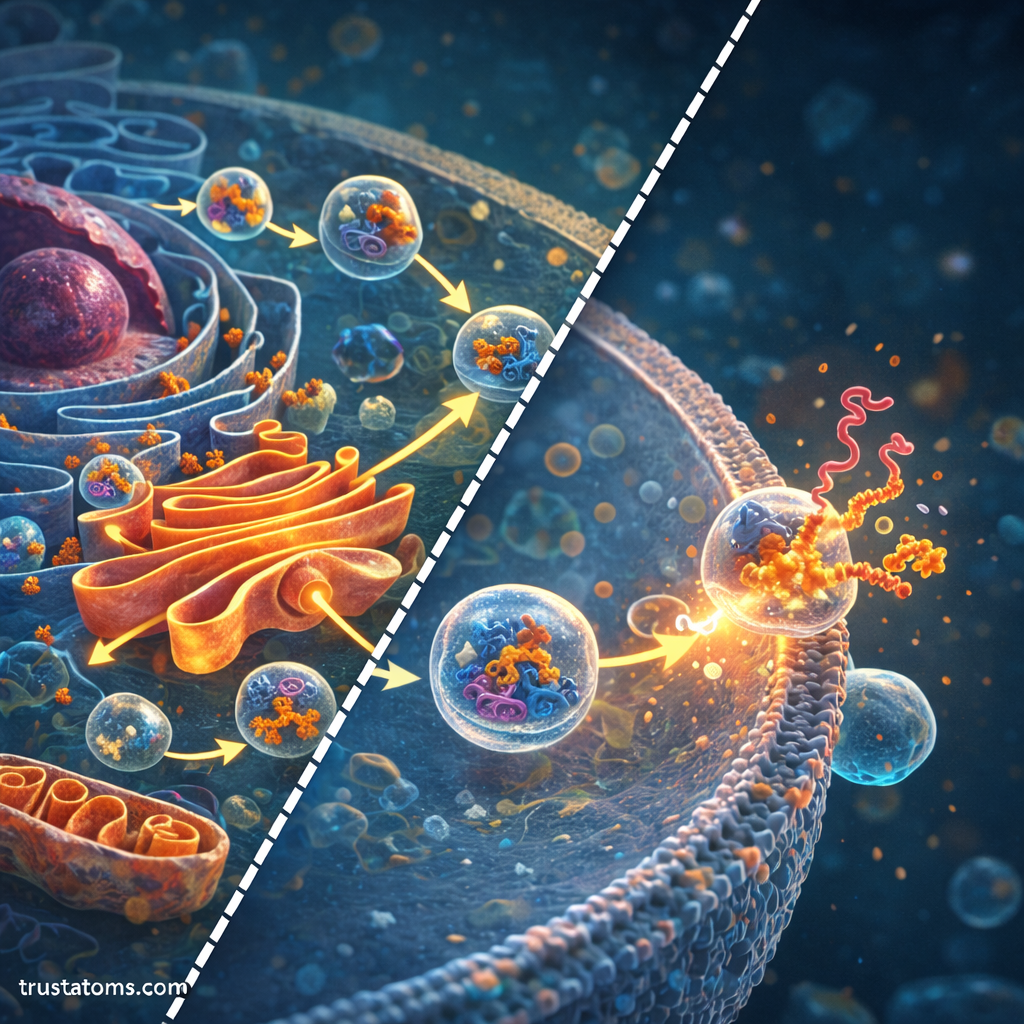
Proteins move between cellular compartments using small membrane-bound structures called vesicles.
Vesicle Formation
Transport vesicles bud off from organelle membranes, carrying proteins to their next destination.
Different vesicle types are involved in various transport steps.
Vesicle Targeting and Fusion
Each vesicle contains molecular markers that guide it to the correct target membrane.
Once the vesicle reaches its destination:
- The vesicle recognizes the target membrane.
- Specialized proteins help the membranes fuse.
- The cargo proteins are delivered to the new compartment.
This highly regulated system ensures proteins are transported accurately.
Major Protein Trafficking Pathways
Cells use several major pathways to transport proteins.
Secretory Pathway
The secretory pathway directs proteins from the ER through the Golgi and eventually to the cell surface or outside the cell.
Proteins following this pathway include:
- Hormones
- Antibodies
- Digestive enzymes
- Membrane receptors
These proteins are packaged into vesicles that fuse with the plasma membrane.
Lysosomal Targeting Pathway
Some proteins are directed to lysosomes, where they help break down cellular waste.
These proteins receive a molecular tag in the Golgi known as a mannose-6-phosphate marker, which directs them to lysosomes.
Nuclear Transport
Proteins destined for the nucleus contain nuclear localization signals.
These proteins pass through specialized channels called nuclear pores, which regulate transport between the cytoplasm and nucleus.
Mitochondrial Import
Proteins that function in mitochondria are synthesized in the cytoplasm and then imported into the organelle.
Special receptor proteins on mitochondrial membranes recognize mitochondrial targeting signals and guide the proteins into the organelle.
Quality Control in Protein Trafficking
Cells maintain strict quality control systems to ensure only properly formed proteins are transported.
Quality control mechanisms include:
- Detection of misfolded proteins
- Degradation through proteasomes
- Recycling of defective molecules
This monitoring system protects cells from potentially harmful protein accumulation.
Protein Trafficking and Human Disease
Disruptions in protein trafficking can cause numerous diseases.
Examples include:
- Cystic fibrosis, caused by improper trafficking of a membrane protein
- Lysosomal storage diseases, resulting from defective enzyme delivery
- Neurodegenerative disorders, linked to protein misfolding and aggregation
Studying protein trafficking helps researchers better understand these conditions and develop new therapeutic strategies.
Advances in Protein Trafficking Research
Modern technologies are providing new insights into how proteins move within cells.
Key research tools include:
- Live-cell imaging
- Fluorescent protein tagging
- Cryo-electron microscopy
- Genetic engineering techniques
These methods allow scientists to observe protein transport processes in real time and uncover previously unknown mechanisms.
Conclusion
Protein trafficking is a fundamental process that ensures proteins reach the correct cellular locations where they can perform their functions. Through a coordinated system involving signal sequences, organelle networks, vesicle transport, and quality control mechanisms, cells maintain precise control over protein distribution.
From the endoplasmic reticulum and Golgi apparatus to specialized organelles and the cell membrane, proteins travel through complex pathways that support cellular metabolism, communication, and structure.
Understanding protein trafficking not only reveals how cells operate but also provides valuable insights into disease mechanisms and potential therapeutic targets in modern medicine.