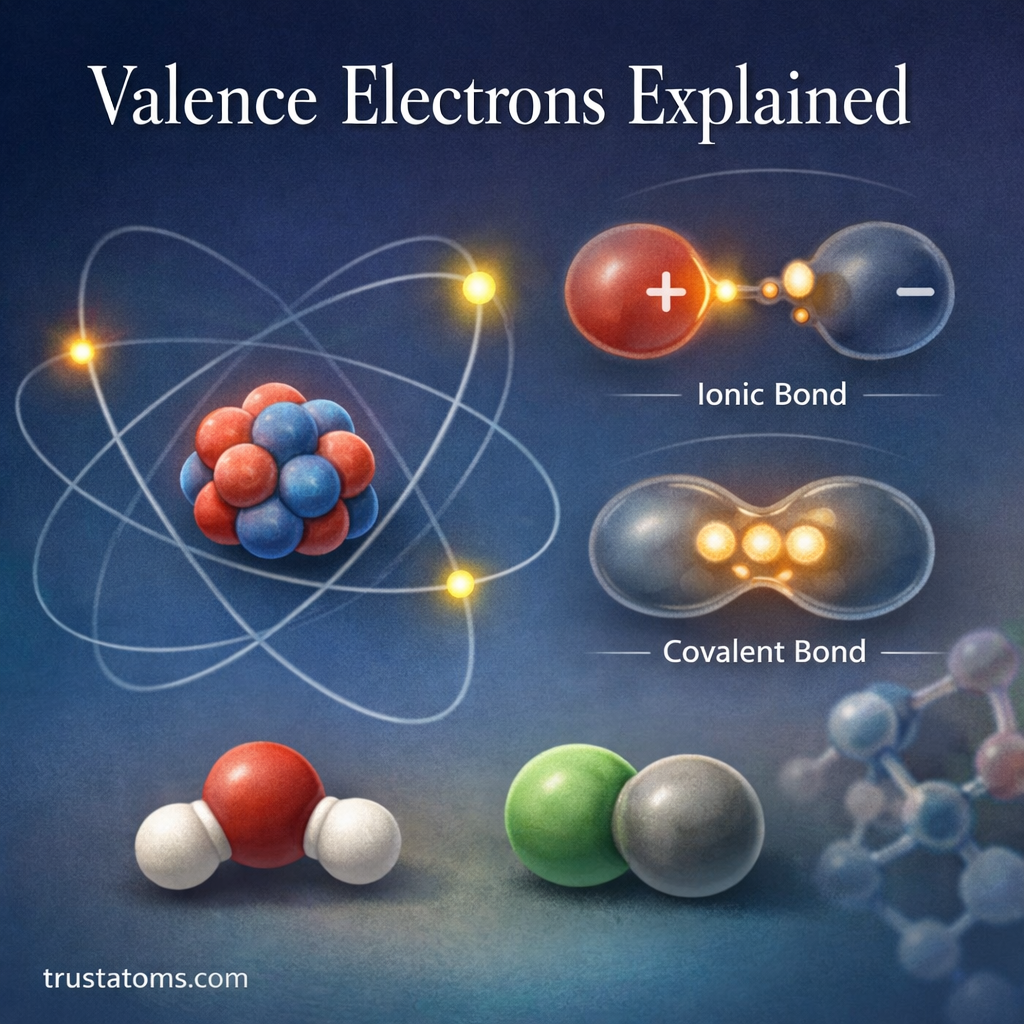
Valence electrons are the electrons located in the outermost shell of an atom. These electrons play a critical role in determining how atoms interact, bond, and form molecules.
In chemistry, understanding valence electrons helps explain why elements behave the way they do, how chemical bonds form, and why certain reactions occur. From the formation of water to the structure of complex molecules, valence electrons are at the center of chemical behavior.
What Are Valence Electrons?
Valence electrons are the electrons found in the outermost energy level (electron shell) of an atom. Because they are the farthest from the nucleus, they are the most likely to participate in chemical bonding.
Atoms naturally seek stable arrangements of electrons. In many cases, this means filling their outer shell with electrons, which is why atoms gain, lose, or share valence electrons when forming chemical bonds.
Key characteristics of valence electrons include:
- Located in the outermost electron shell
- Responsible for chemical bonding
- Determine an element’s chemical reactivity
- Influence the formation of molecules and compounds
Without valence electrons, atoms would not be able to combine to form the vast variety of substances found in nature.
Why Valence Electrons Matter in Chemistry
Valence electrons determine how atoms interact with one another. The number of valence electrons an atom has influences the type of bonds it can form.
Atoms tend to move toward a stable electron configuration, often similar to that of noble gases.
Valence electrons affect several key chemical behaviors:
- How easily an atom reacts with other atoms
- Whether an atom forms ionic or covalent bonds
- The number of bonds an atom can create
- The shape and stability of molecules
For example:
- Sodium has one valence electron and readily loses it.
- Chlorine has seven valence electrons and tends to gain one.
When sodium and chlorine interact, they form sodium chloride (table salt) through the transfer of valence electrons.
The Role of Valence Electrons in Chemical Bonding

Chemical bonds form when atoms interact through their valence electrons.
There are two major types of bonding that involve valence electrons.
Ionic Bonds
Ionic bonds occur when electrons are transferred from one atom to another.
This usually happens between:
- Metals (which lose electrons)
- Nonmetals (which gain electrons)
Example process:
- A metal atom loses one or more valence electrons.
- A nonmetal atom gains those electrons.
- The resulting charged particles attract each other.
This attraction forms an ionic compound.
Covalent Bonds
Covalent bonds occur when atoms share valence electrons.
This commonly happens between nonmetal atoms.
Examples include:
- Water (H₂O)
- Carbon dioxide (CO₂)
- Oxygen gas (O₂)
Sharing electrons allows both atoms to achieve more stable outer electron shells.
How Many Valence Electrons Do Atoms Have?
The number of valence electrons depends on the element’s position in the periodic table.
For main group elements, the group number usually corresponds to the number of valence electrons.
Examples include:
| Periodic Table Group | Valence Electrons | Example Element |
|---|---|---|
| Group 1 | 1 | Sodium |
| Group 2 | 2 | Magnesium |
| Group 13 | 3 | Aluminum |
| Group 14 | 4 | Carbon |
| Group 15 | 5 | Nitrogen |
| Group 16 | 6 | Oxygen |
| Group 17 | 7 | Chlorine |
| Group 18 | 8 | Neon |
Noble gases in Group 18 typically have full outer shells, making them chemically stable and largely unreactive.
The Octet Rule
Many atoms follow a guideline known as the octet rule.
The octet rule states that atoms tend to gain, lose, or share electrons to achieve eight electrons in their outer shell.
This arrangement provides a stable configuration similar to noble gases.
Examples:
- Sodium loses one electron to reach a stable state.
- Chlorine gains one electron to complete its outer shell.
- Oxygen shares electrons in covalent bonds to achieve stability.
While the octet rule explains many chemical behaviors, some elements (especially transition metals) do not always follow it.
How to Determine Valence Electrons
You can determine the number of valence electrons using the periodic table.
Steps to find them:
- Identify the element on the periodic table.
- Locate its group (vertical column).
- For main group elements, the group number indicates the number of valence electrons.
Example:
- Carbon is in Group 14, so it has 4 valence electrons.
- Oxygen is in Group 16, so it has 6 valence electrons.
- Potassium is in Group 1, so it has 1 valence electron.
This information helps predict how atoms will bond and react.
Valence Electrons and Chemical Reactivity
Atoms with nearly empty or nearly full outer shells tend to be the most reactive.
Highly Reactive Elements
Some elements react easily because they are close to achieving a stable electron configuration.
Examples:
- Alkali metals easily lose one valence electron.
- Halogens readily gain one electron.
These strong tendencies make them chemically reactive.
Less Reactive Elements
Elements with full outer shells are far less reactive.
Noble gases such as:
- Neon
- Argon
- Krypton
have complete valence shells, which makes them chemically stable.
Valence Electrons in Everyday Chemistry
Valence electrons are involved in nearly every chemical process around us.
Common examples include:
- Salt forming when sodium and chlorine bond
- Water molecules forming through shared electrons
- Carbon atoms bonding to create organic molecules
- Chemical reactions used in batteries and electronics
From biological systems to industrial chemistry, valence electrons drive the interactions that build matter.
Final Thoughts
Valence electrons are the outermost electrons of an atom and are responsible for chemical bonding and reactivity. By determining how atoms interact, these electrons influence the formation of molecules, compounds, and materials throughout the natural world.
Understanding valence electrons provides a foundation for learning broader chemical concepts such as bonding, reactions, and molecular structure. Whether studying basic chemistry or exploring advanced science topics, valence electrons are a key concept that explains how matter behaves.




