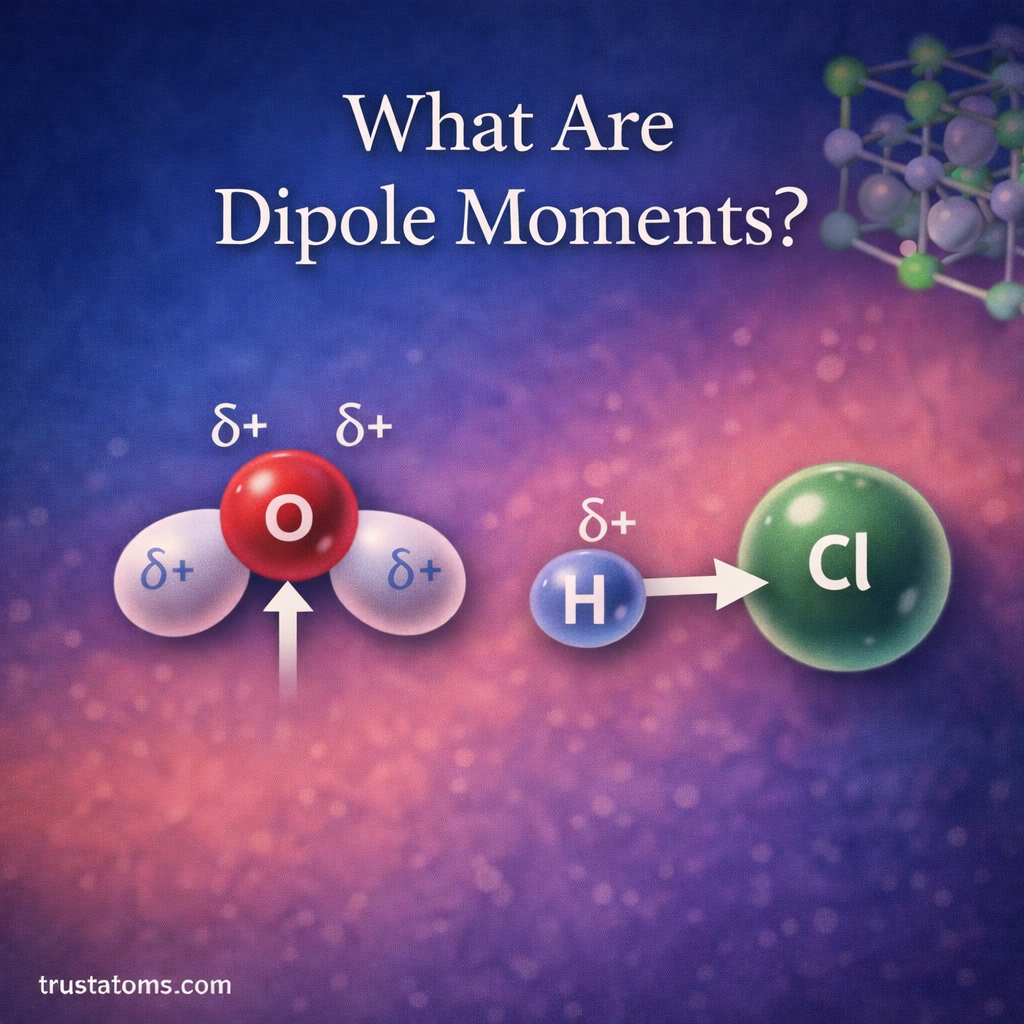
Dipole moments are an important concept in chemistry that describe how electrical charge is distributed within a molecule. When electrons are not shared evenly between atoms, parts of the molecule become slightly positive or slightly negative.
This separation of charge creates a dipole, which affects how molecules interact with each other. Dipole moments help chemists understand molecular polarity, intermolecular forces, solubility, and chemical reactions.
By studying dipole moments, scientists can predict how molecules behave in liquids, gases, biological systems, and chemical reactions.
What Is a Dipole Moment?
A dipole moment is a measure of the separation of positive and negative charges within a molecule.
When electrons are pulled closer to one atom than another, the molecule develops two poles:
- A partially negative region (δ−) where electrons are concentrated
- A partially positive region (δ+) where electrons are less concentrated
The dipole moment describes both:
- The magnitude of the charge separation
- The direction of the polarity
In simple terms, it tells chemists how polar a molecule is.
How Dipole Moments Form
Dipole moments arise from polar covalent bonds.
When atoms with different electronegativities form a bond, one atom pulls electrons more strongly than the other.
This unequal sharing causes:
- Electrons to shift toward the more electronegative atom
- Partial charges to develop on each atom
- A dipole to form along the bond
The direction of the dipole is typically shown with an arrow pointing toward the more electronegative atom.
Factors That Affect Dipole Moments
Several factors influence whether a molecule has a dipole moment and how strong it is.
1. Electronegativity Differences
The larger the electronegativity difference between atoms, the greater the electron displacement.
Greater differences usually create stronger dipole moments.
Example:
- H–Cl has a significant dipole because chlorine strongly attracts electrons.
2. Bond Length
Dipole moment also depends on the distance between charges.
Longer bonds can increase the magnitude of the dipole because the charge separation occurs over a greater distance.
3. Molecular Geometry
Even if individual bonds are polar, the overall dipole moment depends on molecular shape.
Bond dipoles can either:
- Reinforce each other, creating a polar molecule
- Cancel each other out, creating a nonpolar molecule
Geometry plays a major role in determining the final dipole moment.
Dipole Moments and Molecular Polarity
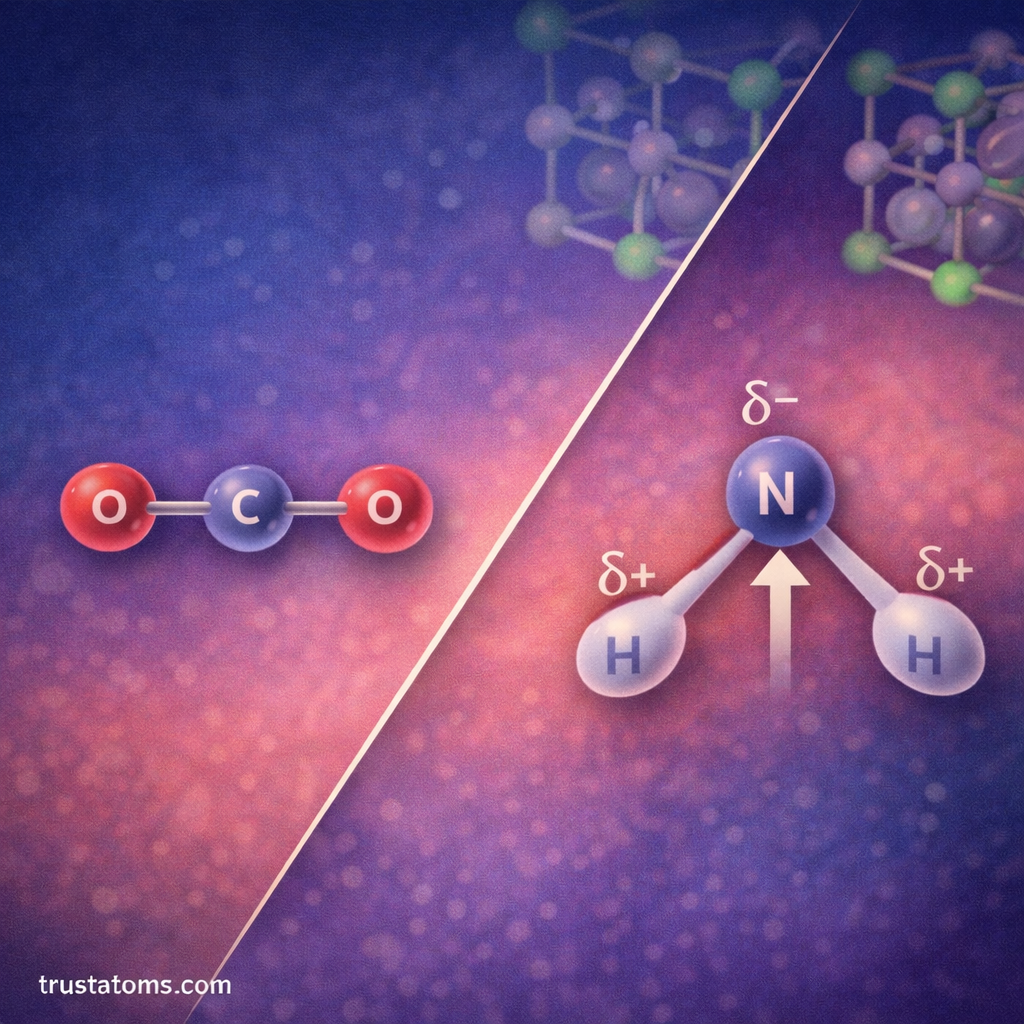
Dipole moments are closely related to molecular polarity.
Polar Molecules
Polar molecules have a net dipole moment because charge distribution is uneven.
Examples include:
- Water (H₂O)
- Ammonia (NH₃)
- Hydrogen chloride (HCl)
These molecules have asymmetrical shapes or uneven electron distribution.
Nonpolar Molecules
Nonpolar molecules have no overall dipole moment.
This happens when:
- Bonds are nonpolar
- Bond dipoles cancel due to molecular symmetry
Examples include:
- Carbon dioxide (CO₂)
- Methane (CH₄)
- Oxygen (O₂)
Even if individual bonds are polar, symmetrical structures can eliminate the overall dipole.
Units Used to Measure Dipole Moments
Dipole moments are measured in a unit called the Debye (D).
The Debye unit reflects the product of:
- The magnitude of the separated charges
- The distance between those charges
Typical dipole moment values include:
- Water: about 1.85 D
- Hydrogen chloride: about 1.08 D
- Carbon dioxide: 0 D (nonpolar molecule)
These values help chemists quantify molecular polarity.
Why Dipole Moments Matter
Dipole moments influence many important chemical properties and behaviors.
1. Intermolecular Forces
Molecules with dipole moments experience dipole–dipole attractions.
These forces influence:
- Boiling points
- Melting points
- Physical state of substances
Stronger dipoles generally lead to stronger intermolecular interactions.
2. Solubility
Dipole moments help explain why certain substances dissolve in others.
Polar molecules dissolve well in polar solvents.
Examples:
- Salt dissolves in water
- Oil does not dissolve in water
This principle is commonly summarized as “like dissolves like.”
3. Chemical Reactivity
Regions of partial positive and negative charge affect how molecules react.
Electrons often move toward:
- Positively charged regions
- Electronegative atoms
Dipole moments help chemists understand reaction mechanisms and molecular interactions.
4. Biological Processes
Dipole moments play a critical role in biological systems.
They influence interactions between:
- Proteins
- DNA molecules
- Enzymes
- Cell membranes
Many biological structures rely on polar interactions to maintain proper shape and function.
How Chemists Represent Dipole Moments
Chemists often show dipole moments using a dipole arrow notation.
This arrow includes:
- A plus sign at the positive end
- An arrow pointing toward the negative end
The arrow shows the direction in which electrons are pulled.
For example:
In hydrogen chloride, the arrow points toward chlorine because it is more electronegative than hydrogen.
Common Examples of Dipole Moments
Water (H₂O)
Water has two polar O–H bonds.
Because of its bent shape, the dipoles do not cancel.
Result:
- Water has a strong dipole moment.
This polarity explains water’s powerful solvent properties.
Ammonia (NH₃)
Nitrogen attracts electrons more strongly than hydrogen.
The molecule’s trigonal pyramidal shape allows the bond dipoles to combine.
Result:
- Ammonia has a net dipole moment.
Carbon Dioxide (CO₂)
Each C–O bond is polar.
However, the molecule is linear, causing dipoles to cancel.
Result:
- CO₂ has no net dipole moment.
Key Takeaways
Dipole moments measure how electrical charge is distributed within molecules.
Important points to remember:
- Dipole moments arise from unequal electron sharing in polar bonds
- They indicate the magnitude and direction of charge separation
- Molecular geometry determines whether dipoles cancel or combine
- Dipole moments influence intermolecular forces, solubility, and chemical behavior
Understanding dipole moments helps chemists predict how molecules interact and behave in chemical reactions, materials, and biological systems.