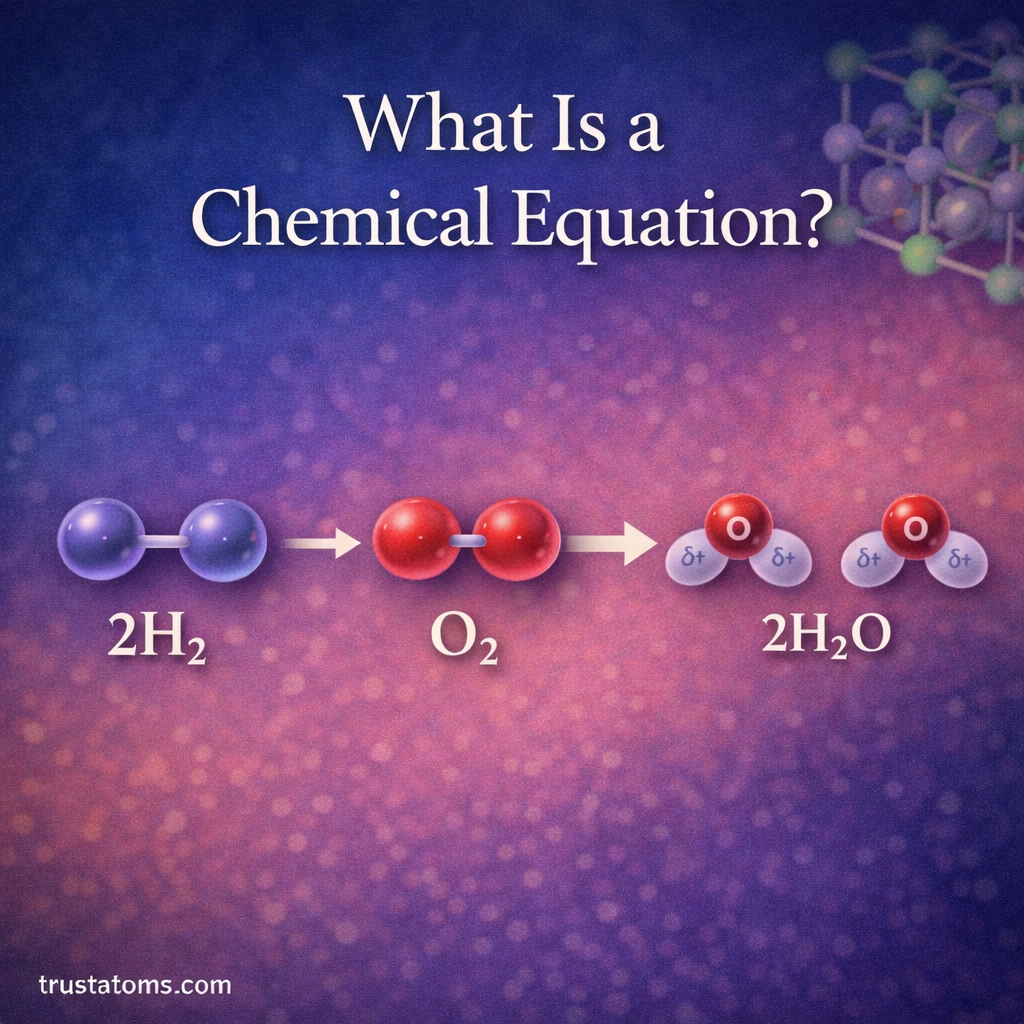
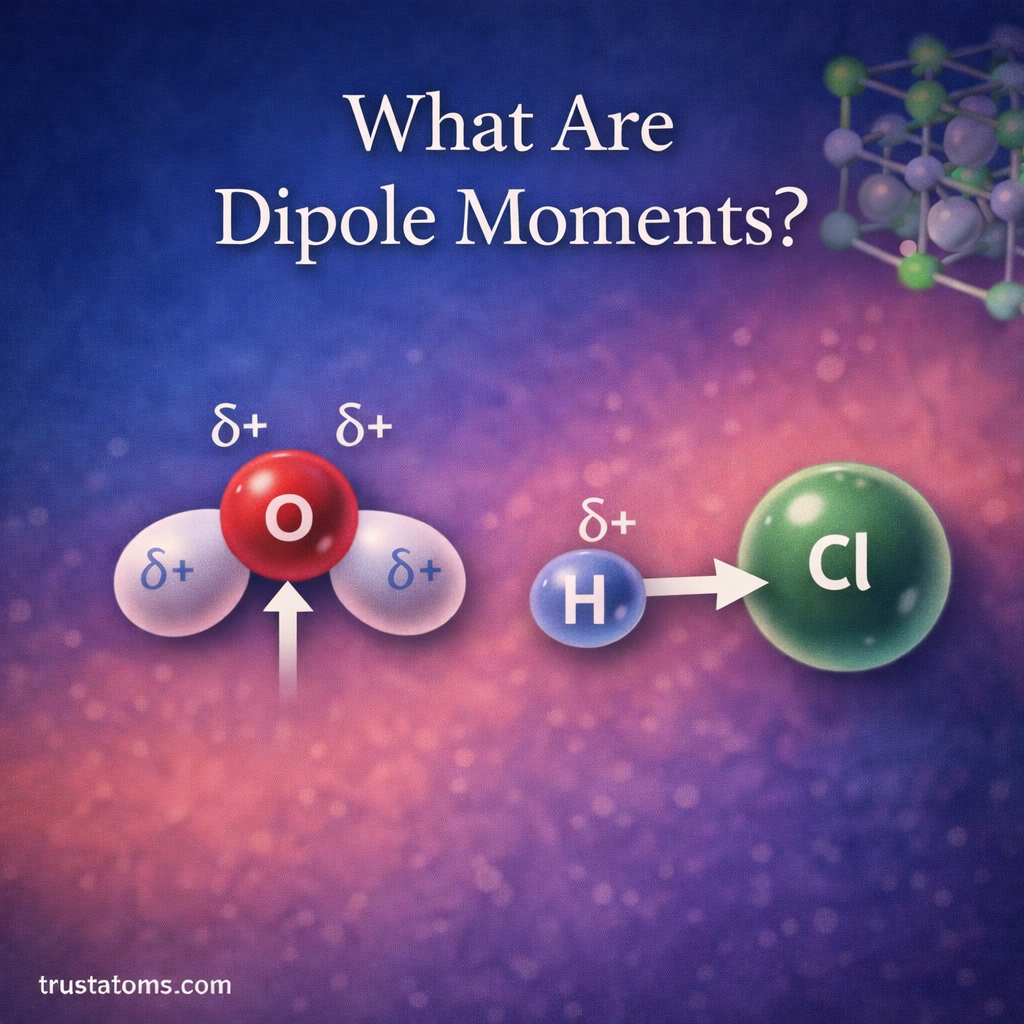
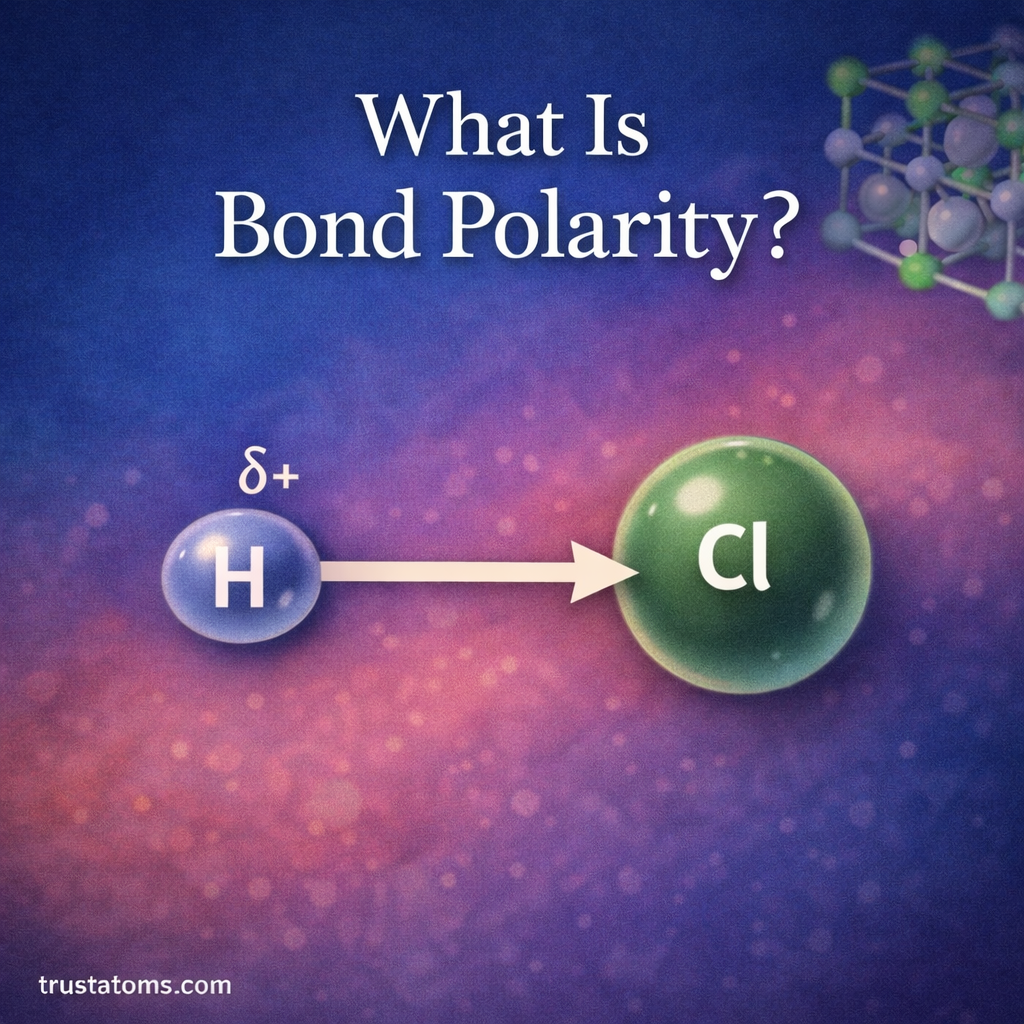
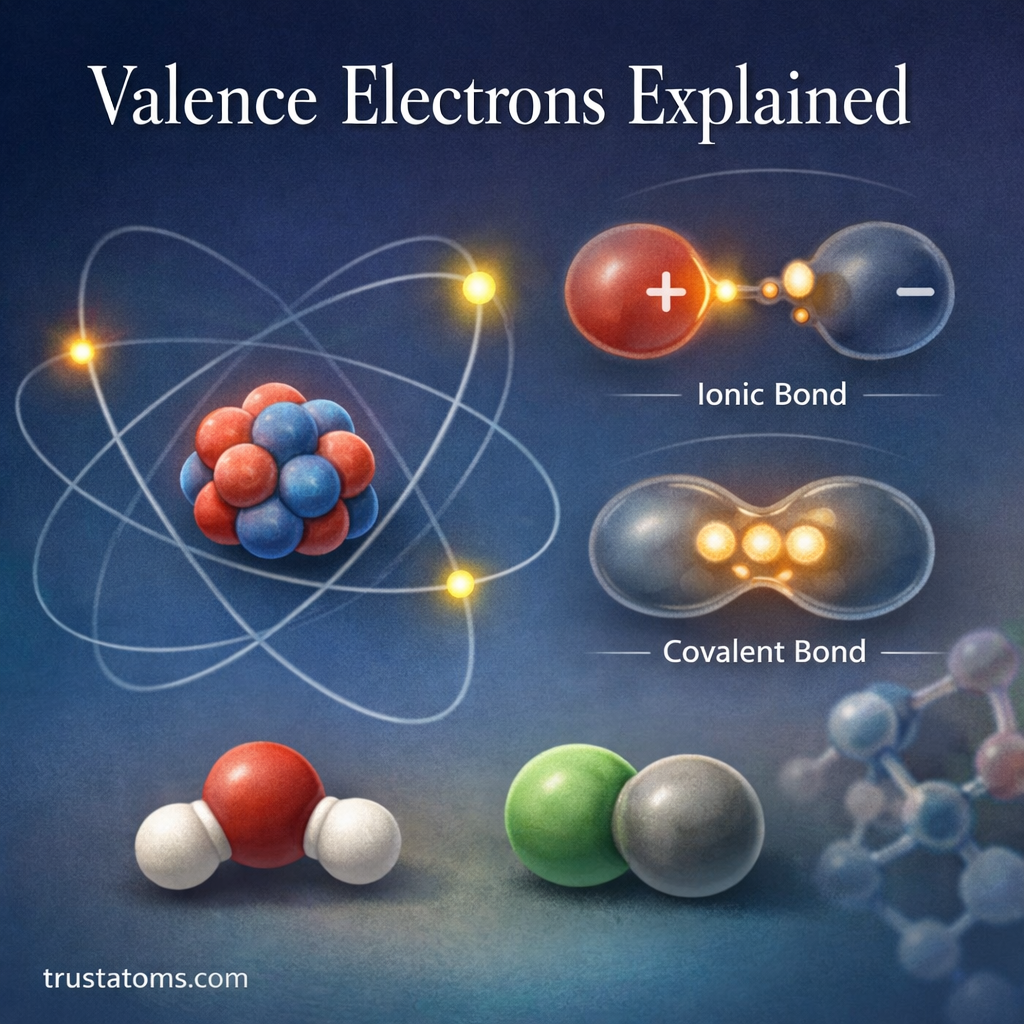
In chemistry, reactions rarely produce the exact amount of product predicted by calculations. Even with carefully controlled experiments, some materials may be lost, reactions may not go to completion, or side reactions may occur. Because of this, chemists use percent yield to measure how efficient a chemical reaction actually is.
Percent yield compares the amount of product obtained in a real experiment to the maximum amount that could theoretically be produced. This value helps scientists determine whether a reaction is working efficiently or if improvements are needed.
Understanding percent yield is an important skill in chemistry, especially when studying reaction efficiency, laboratory experiments, and industrial chemical processes.
Continue reading “What Is Percent Yield?”