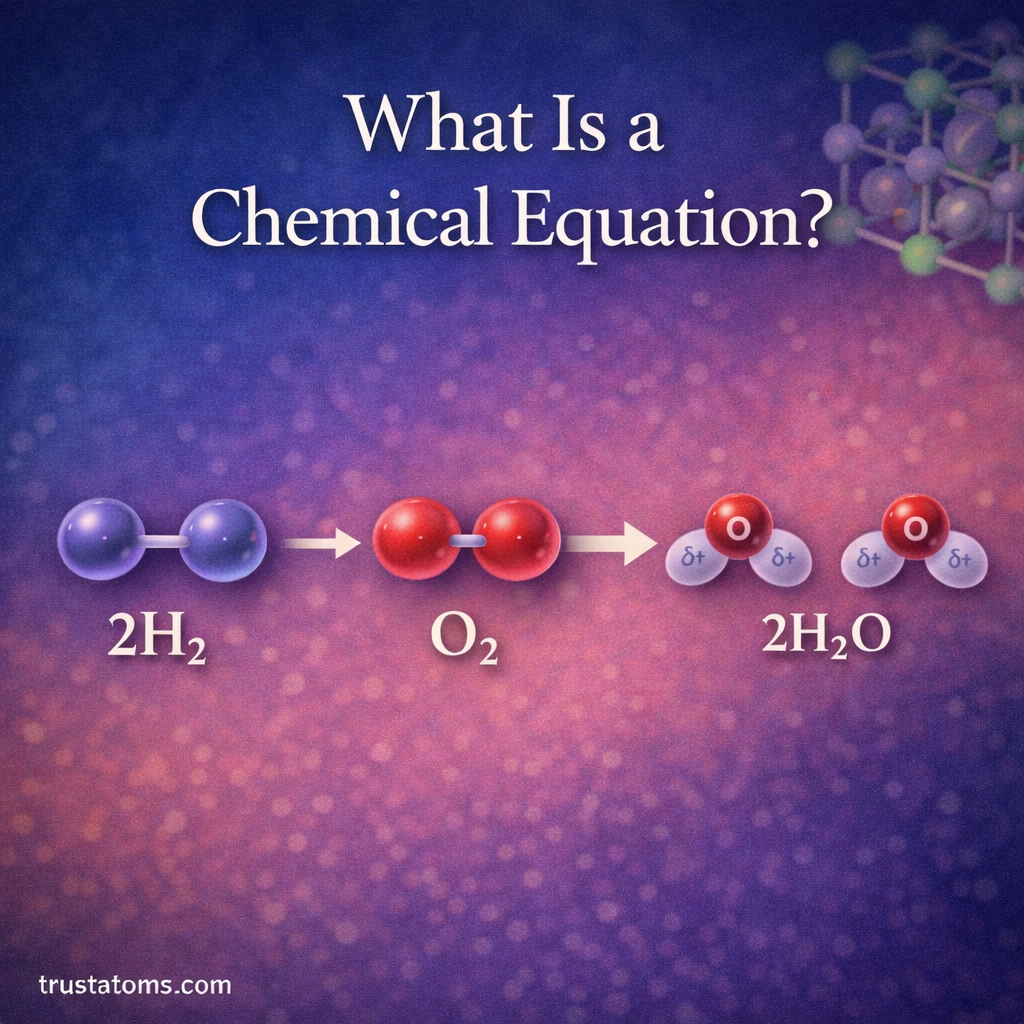
Chemical reactions describe how substances transform into new substances. Chemists represent these reactions using chemical equations, which show the reactants (starting materials) and the products (new substances formed).
However, chemical equations must follow one critical rule: the law of conservation of mass. This means matter cannot be created or destroyed during a reaction. The number of atoms of each element must be the same on both sides of the equation.
Balancing chemical equations ensures that this rule is satisfied and that the equation accurately reflects the reaction taking place.
Continue reading “How to Balance Chemical Equations”